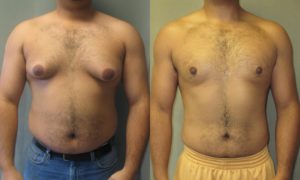
“Let your food be your medicine, and your medicine be your food.” — Hippocrates
Weight Loss:
GLP-1 Receptor Agonists:
-
Semaglutide (Ozempic, Wegovy, Rybelsus)
-
Liraglutide (Saxenda)
- Dulaglutide (Trulicity)
-
Tirzepatide (Mounjaro)
See also:
Introduction
Obesity is a chronic, relapsing disease that places a substantial health burden in affected individuals. This is especially true for those with chronic pain due to the production of inflammatory chemicals by fat cells that contribute significantly to chronic pain. So, excessive fat is not just a mechanical burden contributing to chronic pain but also a chemical burden that drives inflammation and pain.
Guidelines for weight loss and treating obesity encourage lifestyle modifications, including decreased caloric intake, moderate to vigorous exercise, and behavioral therapy as the first steps in the intervention. Achieving and maintaining long-term weight loss is very challenging for many reasons, but largely because of the difficulty of adhering to the necessary lifestyle interventions of diet and exercise.
To gain substantial clinical benefits from weight loss, including reducing cardiovascular risks and pain, it is generally necessary to sustain a weight loss of 5% to 15% of one’s total body weight. However, most anti-obesity medications provide only moderate effectiveness (3%-8% body weight reduction beyond lifestyle interventions alone). Short-term treatment (3-6 months) generally fails to produce either persistent weight loss or long-term health benefits and several agents also have safety concerns.
New treatments that are well-tolerated and able to provide substantial, sustained weight loss over the long term are needed. A new class of drugs, glucagon-like peptide 1 receptor agonists (GLP-1 RA) appear to offer safe and effective weight loss.
Weight Loss Medications
There are currently five FDA-approved medications/classes on the market for weight loss:
- Phentermine-topiramate (Qsymia)
- Orlistat (Xenical),
- Naltrexone-bupropion (Contrave)
- Glucagon-like peptide-1 receptor agonists (GLP-1 R): liraglutide (Saxenda), semaglutide (Ozempic & Wegovy) and dulaglutide (Trulicity)
- Tirzepatide (Mounjaro) – a dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 RA.
GLP-1 Receptor Agonists (GLP-1 RA)
A new class of medications appears to meet these needs, glucagon-like peptide 1 receptor agonists (GLP-1 RA). Weight loss with GLP-1 RAs is thought to be due to improved appetite control with subsequent reduced food intake related also to slowed gastric empying. They are also successful at improving blood sugar (glycemic) control by stimulating insulin secretion and inhibiting glucagon secretion without precipitating hypoglycemia. There are currently two FDA approved GLP-1 RAs: Semaglutide (Ozempic, Welgovy & Rybelsus) and Liraglutide (Saxenda). In addition, there is tirzepatide (Mounjaro) – a dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 RA.
Semaglutide
This review is not intended to dive deeply into semaglutide or other GLP-1 RAs. For links to additional reference articles exploring GLP-1 RAs, click here.
Forms of Semaglutide:
- Injectable: (Welgovy) – FDA-approved for weight loss and Type II diabetes only, not for weight loss
- Oral: (Rybelsus) – FDA-approved for Type II Diabetes only, not for weight loss
- Sublingual: (compounded generic) – Compounded drugs do not come under FDA approval
Injectable Semaglutide: Ozempic & Welgovy
Subcutaneous semaglutide (Ozempic & Welgovy), a GLP-1 receptor agonist FDA approved for adults over 18 y/o with type 2 diabetes, has shown promise for blood sugar control as well as for promoting weight loss in patients both with or without type 2 diabetes. Recent studies using once-weekly 2.4 mg doses of subcutaneous semaglutide for the treatment of overweight/obesity suggest it to be safe and effective.
A recent multi-center, randomized clinical trial published in 2021 evaluated overweight/obese adults who maintained once-weekly treatment for up to 68 weeks with subcutaneous semaglutide. The study subjects experienced sustained weight loss along with significant improvements in waist circumference, systolic blood pressure, and physical functioning.
Weight loss achieved during the study period reached a plateau at week 60 to 68, ultimately resulting in an estimated reduction of 17.4% body weight over the trial. Notably, 86% of participants attained at least 5% weight loss. In contrast, participants who switched back to placebo after 20 weeks gradually regained weight.
Individual counseling sessions are recommended every 4 weeks to help patients adhere to a reduced-calorie diet (500 calorie deficit per day) and increased physical activity is encouraged (i.e. 150 minutes per week of physical activity such as walking). It is suggested that both diet and physical activity be recorded daily in a diary or with use of a smartphone application for review during counseling sessions.
Dosing of Injectable Semaglutide (Ozempic & Welgovy)
Semaglutide, administered with a prefilled pen injector, is initiated at a dose of 0.25 mg once weekly for the first 4 weeks, with the dose increased every 4 weeks until reaching the maintenance dose of 2.4 mg weekly by week 16. In the presence of unacceptable side effects with the 2.4-mg dose, lower maintenance doses are recommended.
While not FDA approved for weight loss, anecdotal evidence supports off-label use of injectable semaglutide to be very helpful for some to lose weight.
Oral Semaglutide: Rebelsus
Rybelsus is a pill form of semaglutide FDA approved for adults over 18 y/o with type 2 diabetes that is to be used along with diet and exercise to improve blood sugar and A1C. Semaglutide is not recommended as the first choice of medicine for treating diabetes type II and is not for use in people with type 1 diabetes.
While not FDA approved for weight loss, anecdotal evidence suggests off-label use of oral semaglutide may be very helpful for some to lose weight.
A 6-month study evaluated 703 adult type 2 diabetics with an average weight of 195 pounds compared daily doses of 7 and 14 mg Rybelsus combined with dietary intervention and exercise. The control group who took a placebo only lost about 3 pounds while some gained weight. Study subjects that that took 7 mg daily lost an average of 5 pounds while those that took 14 mg doses lost an average of 8 pounds.
Dosing of Oral Rybelsus
Rybelsus tablets come in 7 mg or 14 mg strengths. It is taken by mouth on an empty stomach with a sip of plain water (no more than 4 ounces) soon after awakening.
-
-
- After 30 minutes, one can eat, drink, or take other oral medicines For the best effect, it is recommended to eat 30 to 60 minutes after taking semaglutide
- Do not split, crush, or chew semaglutide tablets, it should be swallowed whole
- If one misses a dose of semaglutide, skip the missed dose and take the next dose at the regular time. Do not double the dose to catch up.
- After 30 minutes, one can eat, drink, or take other oral medicines For the best effect, it is recommended to eat 30 to 60 minutes after taking semaglutide
-
Sublingual Semaglutide: Compounded Generic Semaglutide
Due to the overwhelming popularity of what appears to be a very successful means of losing weight, there has been a great demand for semaglutide treatment. Due to the high cost of these medications and lack of insurance coverage for their off-label use, especially for those without diabetes, demand for an affordable version is strong.
Because there is an extremely low absorption of the oral tablets (less than 1% per the labeling for Rybelsus®) and because many patients prefer to avoid injections when possible, a new a patent-pending compounded
formulation of semaglutide for sublingual administration is now available.
Sublingual semaglutide is a liquid form of semaglutide intended for daily use. Although being a compounded version of semaglutide, it is not FDA approved but still recommended only for glycemic control as well as for weight loss in adults over 18 y/o, both with or without type 2 diabetes.
Sublingual semaglutide is to be used along with diet and exercise to improve blood sugar and A1C and facilitate weight loss. Sublingual semaglutide is not recommended as the first choice of medicine for treating diabetes type II and is not for use in people with type 1 diabetes.
More about sublingual semaglutide
This new formulation of sublingual semaglutide is made with Rybelsus tablets and SubMagna SL HMW. SubMagna is an anhydrous, self-emulsifying drug delivery system developed to carry drugs of high molecular weight (HMW) in a sublingual route of administration. This compounding base also has mucoadhesive properties which increase the contact time of the drug to the lining (buccal mucosa) of the sublingual space under the tongue to enhance absorption. The buccal mucosa contains blood and lymphatic vessels which allow drugs to rapidly and directly be absorbed into the systemic blood stream by means of a venous drainage to the superior vena cava. Research has demonstrated that SubMagna SL HMW is able to deliver semaglutide into and through human gingival and oral tissues.
Dosing of sublingual semaglutide
Dosing may be individualized based on response to the medication, including side effects and/or effectiveness. The medication should be taken at least 30 min before eating and waiting at least 90 minutes before swallowing.
-
-
- First Month: Place 0.25 ml (0.5 mg) once daily for 1 week, then 0.5 ml (1 mg) once daily thereafter
- Second Month: Place 0.5 ml (1 mg) once daily
- Third Month: Place 0.75 ml (1.5 mg) once daily
- Fourth Month: Place 1 ml (2 mg) once daily therafter
-
Cost of sublingual semaglutide
The estimated cost for 15 ml of sublingual semaglutide is $185. This amount of 15 ml/month is ample for the first two months but the third and fourth month dose increases may require up to 30 ml, or up to $370/month.
Maintenance of Treatment
Similar to trials of other anti-obesity medications that emphasize the chronicity of obesity, studies of semaglutide identified the need for continuous, prolonged weekly treatment with semaglutide in order to maintain maximal weight loss. It is not recommended as a short term treatment for rapid weight loss.
The significant and sustained weight loss demonstrated with continued semaglutide treatment has also been accompanied by improvement in waist circumference, lipid profiles, and glucose metabolism, all of which are significant cardiovascular risk factors. Sustained weight loss of this magnitude has been shown to improve obesity-related complications including type 2 diabetes.
In patients with diabetes type 2 who are at high risk for cardiovascular disease, semaglutide not only has demonstrated improvement in blood sugar and body weight but it has also been shown to lower the rate of cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke. The superiority of semaglutide in reducing body weight compared with other anti-diabetic medications has been relatively well established by clinical trials.
Possible side effects of semaglutide
Typically with the class of GLP-1 receptor agonists, the most frequently reported adverse events are transient, mild to moderate in severity and gastrointestinal in nature (namely nausea and vomiting, constipation and abdominal pain). These symptoms usually occur only during the initial period when semaglutide is escalated to the target dose. A common effect of GLP-1 receptor agonists is decreased appetite, the primary mechanism for their effectiveness in weight loss. Over one trial period, only a few participants (5.3%) discontinued treatment because of adverse events and those who continued treatment beyond 20 weeks were unlikely to experience significant problems thereafter.
Precautions
Semaglutide should be used with caution in those who:
-
- Have or have had problems with their pancreas or kidneys
- Have a history of vision problems related to diabetes
- Are pregnant or plan to become pregnant. It is not known if semaglutide is harmful to an unborn baby therefore one should stop taking semaglutide two months before planning to become pregnant or who are breastfeeding or plan to breastfeed. Breastfeeding is not recommended while taking semaglutide.
- Drink alcohol. Use of alcohol should be cautious and limited while using semaglutide because it can increase the risk of developing low blood sugar
Side effects of semaglutide compared with other weight loss medications
Phentermine-topiramate has been associated with severe cardiovascular outcomes, while orlistat use increases the risk of renal failure and hepatotoxicity. Semaglutide, on the other hand, lowered death from cardiovascular causes by 26% when compared with placebo. Semaglutide does not affect overall renal function and is considered safe for use in patients with mild to moderate renal impairment. Based on these safety findings, semaglutide appears to be a better alternative to other anti-obesity medications currently on the market. It should also be noted that, semaglutide (like other GLP-1 analogs) has a low risk of hypoglycemia.
Potential for serious side effects with semaglutide
Most people using semaglutide do not have serious side effects, however, they may include:
-
- Possible thyroid tumors, including cancer: In studies with rodents, semaglutide caused thyroid tumors, including thyroid cancer but it is not known if semaglutide causes thyroid tumors or cancer in humans. Therefore, individuals with a personal or family history of thyroid cancer or an endocrine system condition called Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) should not take semaglutide.
- Inflammation of the pancreas (pancreatitis): Pancreatitis may be associated with severe pain in the abdomen that usually persists, with or without vomiting, and may radiate into the back. Based on findings from animal studies, treatment with GLP-1 RAs is contraindicated in patients with a history of chronic or idiopathic acute pancreatitis.
- Changes in vision: Changes in vision during treatment with semaglutide can occur. A higher incidence of retinopathy complications, including vitreous hemorrhage and blindness, have been reported in patients treated with semaglutide compared with placebo. This is believed to be related to the rapidity and magnitude of glycemic improvement rather than a direct side effect of semaglutide.
- Low blood sugar (hypoglycemia): Treatment with semaglutide can cause a drop in blood sugar that may become symptomatic, especially if taken with another medicine that can cause low blood sugar, such as a sulfonylurea or insulin. Drinking large quantities of alcohol, not getting enough calories from food, or doing unusually heavy exercise may also lead to low blood sugar. Signs and symptoms of low blood sugar may include: dizziness or lightheadedness, blurred vision, anxiety, irritability or mood changes, sweating, slurred speech, hunger, confusion or drowsiness, shakiness, weakness, headache, fast heartbeat, and feeling jittery. If symptoms occur, one can rapidly raise their blood sugar by eating a quick source of sugar such as table sugar, honey, candy, or drink fruit juice or non-diet soda.
- Kidney impairment or kidney failure: In the presence of diarrhea and/or vomiting, people who have kidney impairment may become dehydrated which may worsen kidney function. GLP-1 RAs are contraindicated in those with severely impaired renal function.
- Serious allergic reactions: Serious allergic reactions to GLP-1 RAs is rare. Symptoms of serious allergic reactions, include swelling of the face, lips, tongue, or throat; problems breathing or swallowing; severe rash or itching; fainting or feeling dizzy; or very rapid heartbeat.
- Gallbladder problems. Gallbladder problems, principally gall stones, ranges in studies between 0.2% and 4.9% in people who take semaglutide. Symptoms of gallbladder problems may include pain in the upper right or mid-abdomen, yellowing of skin or eyes (jaundice), fever, and clay-colored stools.
- Cardiovascular issues, including tachycardia and arrhythmias, ranged between 1.5% and 9.8% in STEP trials 1–4.
If these symptoms occur, one should call their healthcare provider right away or go to the emergency room
Drug Interactions:
Drug interactions can change how medications work and may increase the risk for serious side effects.
Beta-blocker medications (such as metoprolol, propranolol, or glaucoma eye drops such as timolol) may prevent the fast, pounding heartbeat one would usually feel when their blood sugar falls too low (hypoglycemia). However, other symptoms of low blood sugar, such as dizziness, hunger, or sweating, are not affected by these drugs.
Many drugs can affect blood sugar, so any change any medications should be reviewed by a doctor or pharmacist as to how the medications may affect blood sugar. Blood sugar may need to be checked regularly and shared with a doctor.
.
Liraglutide (Saxenda)
Another GLP-1 receptor agonist, liraglutide, is also approved for weight management at a 3 mg/day dosage. A 56 week study with liraglutide demonstrated weight losses of 10% body weight in 33% of subjects and 15% body weight of subjects in 14% of subjects. In clinical trials, weight loss with liraglutide was of lesser magnitude and appeared to plateau earlier (at 20 or 40 weeks) compared with semaglutide (plateaus at 60-68 weeks).
A 2022 systematic review of the effects of semaglutide and liraglutide in individuals with obesity or overweight without diabetes concluded that liraglutide and semaglutide therapy both led to clinically relevant (⩾5%) weight loss in 48.2%–88.7% of obese/overweight adults without diabetes. Furthermore, both liraglutide and semaglutide were well-tolerated.
Liraglutide is structurally highly similar to semaglutide with only a few chemical modifications. The once-daily dosing regimen of 3.0 mg liraglutide has been approved by the FDA for the treatment of obesity. However, the once-daily injection regimen can cause physical and financial stress for some adults, so the once-weekly injection model for the GLP-1RAs has been implemented by some.
One study directly compared liraglutide and semaglutide and concluded that semaglutide 0.2 mg once-daily showed superior weight loss compared to liraglutide 3.0 mg once-daily. Other studies also found that semaglutide is more effective in reducing body weight than liraglutide; however, the vast majority of the studies were based on patients with diabetes and weight loss was not the main primary outcome.
Conclusions
Based on the effectiveness of these FDA approved GLP-1 receptor agonists, they may offer pharmacological options for overweight and particularly obese patients who might be considering bariatric surgery, which is currently considered the most effective and reliable intervention available for weight management. In comparism, 40% of participants in the semaglutide study trial who took semaglutide for 68 weeks lost 20% or more of their body weight, which approached the level of weight loss seen with sleeve gastrectomy.
It remains important to combine lifestyle modifications such as dietary and physical activity in the treatment of obesity. Dietary modification is fundamental for the long-term management of obesity and includes a variety of dietary interventions, including intermittent fasting, calorie restriction and different macronutrient compositions (e.g. low-fat diets, low-carbohydrates), avoidance of special foods, and following a certain dietary pattern (e.g. Mediterranean diet).
Physical activity is also crucial for successful weight loss, as it can induce energy deficit and preserve lean muscle mass. The American College of Sports Medicine (ACSM) recommends that individuals exercise 150–250 minutes/week, > 150–250 minutes/week, and 200–300 minutes/week to prevent weight gain, achieve weight loss, and maintain weight loss, respectively. Another clinical trial showed that exercise combined with liraglutide was approximately twice as effective as either treatment alone in reducing body weight and body-fat percentage. It was also associated with the improvements in insulin sensitivity, cardiorespiratory fitness, and maintaining a good mood.
The optimal treatment for obesity should be individualized where treatment decisions consider age, coexisting diseases, drug tolerance, and economic and local medical conditions, For example, naltrexone/ bupropion should not be The ideal treatment for obesity should be a highly individualized, personalized medicine. Treatment decisions will consider age, coexisting diseases, drug tolerance, and economic and local medical conditions. For example, naltrexone/ bupropion does not apply to uncontrolled hypertension patients and phentermine/topiramate treated patients should be monitored closely for depression. However, there is no consensus or guidelines to help clinicians make decisions on which drugs to choose.
Tirzepatide (Mounjaro)
Tirzepatide (Mounjaro) is an injectable prescription medication that is used along with diet and exercise to improve blood sugar (glucose) in adults with type 2 diabetes mellitus. It is a dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 RA that is FDA-approved for Type II diabetes only, not for weight loss. Mounjaro is dosed in once-weekly, subcutaneous injection dose-escalations, starting with 5 mg, then 10 mg, then up to 15 mg doses over 20 weeks.
Weight Loss with Tirzepatide
A 2022 study evaluated the use of Mounjaro in subjects with a mean body weight of 231 lbs, and a mean BMI of 38 at baseline, in which 94.5% of participants had a BMI of 30 or higher. The mean percentage weight loss after 72 weeks was 15%, with 5-mg weekly doses of tirzepatide, 19.5% with 10-mg doses, and 20.9% with 15-mg doses compared to 3.1% with placebo.
The percentage of participants who had weight loss of 5% or more was 85%, 89% and 91% with 5 mg, 10 mg, and 15 mg doses of tirzepatide respectively, compared to 35% with placebo. The percentage of participants who had weight loss of 20% or more was 50% and 57% of participants in the 10-mg and 15-mg groups, as compared with 3% in the placebo group.
Side Effects with Tirzepatide
The most common side effects with tirzepatide are nausea, vomiting and diarrhea, mild to moderate in severity, occurring primarily during dose escalation. Side effects cause treatment discontinuation in 4.3%, 7.1%, 6.2%, of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses respectively compared to 2.6% with placebo. Hypoglycemia has been reported in <2% of patients taking tirzepatide.
Tirzepatide vs Semaglutide
A 2021 study comparing tirzepatide and semaglutide concluded that tirzepatide at all doses was superior to semaglutide with weight reductions and improvement of hemoglobin A1c greater with tirzepatide than with semaglutide.
Resources
References
Appetite Suppressants
Appetite Suppressants (OTC) – Alternative (CAM)
Appetite Suppressants – Prescription Medications (Rx) List
- Adipex (Phentermine)
- Belviq (Lorcaserin)
- Bupropion
- Contrave (Bupropion/Naltrexone)
- Invokana (Canagliflozin)
- Naltrexone
- Orlistat (Xenical, Alli)
- Qsymia (Phentermine and Topiramate)
- Saxenda (Liraglutide)
- Topamax (Topiramate)
- Wegovy (Semaglutide)
Appetite Suppressants (Rx) – Semaglutide
- Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity – PMC – 2021
- Review Wegovy (semaglutide)- a new weight loss drug for chronic weight management – 2022
- Once-Weekly Semaglutide in Adults with Overweight or Obesity – 2021
- Once-Weekly Semaglutide in Adults with Overweight or Obesity – PubMed – 2021
- Effect of semaglutide and liraglutide in individuals with obesity or overweight without diabetes- a systematic review – 2022
- GLP-1-Medications-Article-References-and-PubMed-Links
- Sublingual Semaglutide Supportive deposition
- The Impact of GLP1 Agonists on Bone Metabolism: A Systematic Review.- 2022
- Efficacy and safety of dulaglutide 3.0 and 4.5 mg in patients aged younger than 65 and 65 years or older: Post hoc analysis of the AWARD‐11 trial – 2021
Appetite Suppressants (Rx) – Tirzepatide (Mounjaro)
- Tirzepatide Once Weekly for the Treatment of Obesity – PubMed – 2022
- Effects of Tirzepatide, a Dual GIP and GLP-1 RA, on Lipid and Metabolite Profiles in Subjects With Type 2 Diabetes – PubMed – 2022
- Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes – PubMed – 2021
Weight Loss Program
Weight Loss Program – Body Composition Analysis (BCA)
- Body Composition Analysis – Patient Preparation handout
- Measurement of visceral fat by abdominal bioelectrical impedance analysis is beneficial in medical checkup. – PubMed – NCBI
- The clinical importance of visceral adiposity – a critical review of methods for visceral adipose tissue analysis – 2012
- The use of bioelectrical impedance to detect excess visceral and subcutaneous fat – 2007
Weight Loss Program – NLAL Lipolysis
NLAL Lipolysis – Handouts
NLAL Lipolysis – References
- Fat Liquefaction – Effect of Low-Level Laser Energy on Adipose Tissue
- Application of low-level laser therapy for noninvasive body contouring. – PubMed – NCBI
- Body contouring using 635-nm low level laser therapy. – PubMed – NCBI
- Efficacy of low-level laser therapy for body contouring and spot fat reduction. – PubMed – NCBI
- Independent evaluation of low-level laser therapy at 635 nm for non-invasive body contouring of the waist, hips, and thighs. – PubMed – NCBI
- Low-level laser therapy as a non-invasive approach for body contouring: a randomized, controlled study. – PubMed – NCBI
Foods
Weight Loss
CAM – Supplements for weight loss:
Eating Behavior
Eating Behavior – Emotional Eating
Eating Behavior – ExternalEating Cues
Eating Behavior – Fasting
- Calorie restriction increases life span: a molecular mechanism. – PubMed – NCBICalorie restriction increases muscle mitochondrial biogenesis in healthy humans. – 2007
- A double-blind, placebo-controlled test of 2 d of calorie deprivation – effects on cognition, activity, sleep, and interstitial glucose concentrations – 2008
- Fasting and refeeding differentially regulate NLRP3 inflammasome activation in human subjects – 2015
Maladaptive Eating
Maladaptive Eating – Cravings
Cravings – Overview
-
- Food craving – new contributions on its assessment, moderators, and consequences – 2015
- Pickles and ice cream! Food cravings in pregnancy – 2014
- Relationship of cravings with weight loss and hunger – Results from a 6 month worksite weight loss intervention – 2013
- How Relevant is Food Craving to Obesity and Its Treatment? – 2014
Cravings – Treatment
Cravings Treatment – CAM Supplements
Maladaptive Eating – Binge Eating Disoder (BED)
BED – Overview
BED – Dopamine </sp an>
BED – Treatment
BED – Treatment, Overview
BED -Treatment, Bupropion
BED -Treatment, Chromium
-
- Chromium Picolinate – Summary
- A Double-blind, Randomized Pilot Trial of Chromium Picolinate for Binge Eating Disorder – Results of the Binge Eating and Chromium (BEACh) Study – 2013
- Dietary chromium supplementation for targeted treatment of diabetes patients with comorbid depression and binge eating. – PubMed – NCBI
BED -Treatment, Contrave
Obesity
Obesity – Visceral Fat
Obesity, Visceral Fat – Overviews
Obesity, Visceral Fat – Biomarkers
Obesity, Visceral Fat Biomarkers – Adinopectin
Obesity, Visceral Fat Biomarkers – IL-6
Obesity, Visceral Fat Biomarkers – Hypertriglyceridemia
Obesity, Visceral Fat – Exercise
Obesity, Visceral Fat – Genetics
Obesity, Visceral Fat – Stress
Obesity – Vitamins
Obesity, Vitamins – Overviews
Obesity – Vitamin C
Obesity – Vitamin D
-
- Decreased bioavailability of vitamin D in obesity – 2000
- Higher visceral fat area increases the risk of vitamin D insufficiency and defic
iency in Chinese adults – 2015 - Obesity and vitamin D 2004
- Rising serum 25-hydroxy-vitamin D levels after weight loss in obese women correlate with improvement in insulin resistance. – PubMed – NCBI
- The Effect Of Vitamin D Supplementation On Serum 25OHD In Thin And Obese Women – 2012
- The Longitudinal Association of Vitamin D Serum Concentrations & Adiposity Phenotype – 2013
- Vitamin D and Its Relationship with Obesity and Muscle – 2014
- Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers – 2009
- Update on vitamin D – pros and cons – 2015
Obesity – Comorbid Conditions
Obesity – Fatty Liver
Obesity – Kidney Disease
Obesity – Metabolic Syndrome
-
- Association of visceral fat area with chronic kidney disease and metabolic syndrome risk in the general population
- Abdominal Obesity and the Metabolic Syndrome – Contribution to Global Cardiometabolic Risk – 2008
- Metabolic syndrome and adipose tissue: new clinical aspects and therapeutic targets. – PubMed – NCBI
- Subcutaneous and Visceral Adipose Tissue: Their Relation to the Metabolic Syndrome: Endocrine Reviews: Vol 21, No 6
Obesity – Oxidative Stress
Obesity – Sleep Apnea
Emphasis on Education
Accurate Clinic promotes patient education as the foundation of it’s medical care. In Dr. Ehlenberger’s integrative approach to patient care, including conventional and complementary and alternative medical (CAM) treatments, he may encourage or provide advice about the use of supplements. However, the specifics of choice of supplement, dosing and duration of treatment should be individualized through discussion with Dr. Ehlenberger. The following information and reference articles are presented to provide the reader with some of the latest research to facilitate evidence-based, informed decisions regarding the use of conventional as well as CAM treatments.
For medical-legal reasons, access to these links is limited to patients enrolled in an Accurate Clinic medical program.
Should you wish more information regarding any of the subjects listed – or not listed – here, please contact Dr. Ehlenberger. He has literally thous
ands of published articles to share on hundreds of topics associated with pain management, weight loss, nutrition, addiction recovery and emergency medicine. It would take years for you to read them, as it did him.
For more information, please contact Accurate Clinic.
Supplements recommended by Dr. Ehlenberger may be purchased commercially online or at Accurate Clinic.
Please read about our statement regarding the sale of products recommended by Dr. Ehlenberger.
Accurate Supplement Prices
.
