“Despair is suffering without meaning” – Viktor Frankl
Acute Pain: Avoiding Transition to Chronic Pain
The “normal” course of pain is one that resolves as injured tissues heal. Unfortunately, as often occurs in the case of pain arising from acute neck, back or joint injuries – even pain arising from surgery – these pains may not resolve despite the apparent healing of injuries. Instead, sometimes acute post-injury pain becomes chronic and leads to a significant impairment of quality of life. As such, when treating acute pain it is important to take measures to reduce the likelihood and/or severity of the chronification of pain.
Moving beyond mere pain management to total pain recovery
This educational section is directed at understanding and reviewing what can be done to reduce the risk and/or severity of acute pain transitioning to chronic pain. For a general review of the management of acute post-injury pain, see Acute Pain: Initial Post-Injury.
For a condensed overview of treatment options for reducing the transition of acute to chronic pain, click here.
See also:
- Acute Pain: Initial Post-Injury
- The Transition of Acute Post-Injury Opioid Use to Chronic Opioid Use
- Central Sensitization
- Neuroinflammation
- Neurobiology of Pain
- Traumatic Brain Injury (TBI)
See also:
see also:
Definitions and Terms Related to Pain
Key to Links:
- Grey text – handout
- Red text – another page on this website
- Blue text – Journal publication
Goals
There are four primary goals in the management of acute post-injury pain:
(1) Reduce the severity and duration of the acute post-injury pain
(2) Reduce the risk of the acute post-injury pain from becoming chronic
(3) Enhance the individual’s coping skill relative to living with pain
(4) Facilitate improved engagement in daily activities and reduce functional impairment.
The Transition of Acute to Chronic Post-Injury Pain
While the occurence of acute pain in the post-injury period is to be expected by most patients, the transition of acute post-injury pain to become a chronic, possibly life-long source of pain is generally not expected. Poorly managed acute pain increases the risk of chronic pain which is defined as pain lasting for more than 3 months. One year after traumatic injury nearly two-thirds of patients report at least moderate severity pain and three out of four patients report that pain interferes with their activities of daily living, including social engagement, work, and cognitive and emotional functioning. Persisting pain is associated with decreased self-efficacy and increased risk of depression which can be even more pronounced after trauma. Traumatic events cause distress that can exacerbate pain and trigger a trauma-pain-distress feedback loop.
The most significant, consistent factor contributing to the risk of progression of acute to chronic post-injury pain is the duration of severe acute post-injury pain. The general management of acute post-injury pain is detailed in the educational section: Acute Pain: Initial Post-Injury. In this section, the emphasis is on reducing the transition of acute to severe chronic pain and will focus on measures that can be taken in addition to those taken to manage acute pain post-injury..
Chronic Post-Surgical pain (CPSP)
Much of the research done to evaluate the transition from acute to chronic pain has been done looking at post-surgical pain (See: Post-Surgical Pain). Surgically-induced chronic pain is a significant clinical problem, with persistent pain estimated to occur in 10–50% of individuals after common operations. Specific surgeries have been associated with higher incidences of chronic post-surgical pain (CPSP), such as mastectomy, thoracotomy (open chest surgery) and inguinal hernia repair. Some patients develop their CPSP almost immediately after surgery, while in others it starts weeks or months after the surgery.
Risk Factors for the Transition of Acute to Chronic Post-Injury Pain
Although chronic pain often begins with an acute injury, it can become a new and self-sustaining disease in patients with genetic, emotional, or socioeconomic predisposition. This transition is most common in patients with direct injury to nerves or with complex injuries that are slow to heal, but it can also occur after relatively trivial injuries in otherwise healthy patients.
There is marked patient variability in the response to injury, so that not all injuries lead to chronic pain. A number of factors related to the individual patient may increase the risk of developing chronic post-injury pain include duration and severity of acute post-injury pain, preexisting pain, age, genetics and psychological variables.
Duration and Severity of Acute Post-Injury Pain
As noted above, one of the most significant, consistent factors that contributes to the progression of acute to chronic post-injury pain is the duration of severe acute post-injury pain. This is why it is so important to treat acute post-injury pain aggressively in hopes of avoiding the central sensitization (see below) that contributes to the evolution of chronic pain. Furthermore, one must understand that studies of musculoskeletal injuries, including ankle sprains and fractures, have found no correlation between perceived pain intensity and the degree of injury severity. Degrees of pain intensity and the magnitude of perceived limitations are better accounted for by the individual’s psychosocial aspects of illness than by measures of pathophysiology.
Pre-Injury Chronic Pain
As discovered in the study of post-operative pain, the presence of pre-operative pain, regardless of the relationship to surgical site, significantly increases the risk of developing chronic post-surgical pain. This finding likely can be extrapolated to post-injury pain. A pre-existing chronic pain condition is believed to sensitize the brain and escalate the process of developing chronic post-injury pain (central sensitization). Multiple spinal and brain regions have been found to be modified by chronic pain including dorsal horn neurons in the spinal cord and regions not only involved in sensory and pain processing (thalamus, and posterior insula) but also regions involved in emotional processing and subjective magnitude assessment of pain (cingulate cortex, insula, periaqueductal grey, ACC, prefrontal cortex).
In other words, the changes in the brain associated with pre-injury chronic pain establishes a contextual setting in which the pain associated with new traumatic pain may be magnified and chronified.
Age
Adult nervous systems, especially in those over 65, are less adaptive and more likely to give rise to chronic pain while chronic post-injury pain is relatively uncommon in children. However, surgeries at an early age may have long-lasting consequences on subsequent sensitivity to pain. Gender studies have not identified clear cut differences between men and women.
Genetics
Data from research clearly suggest a genetic predisposition for those who develop chronic pain following a precipitating incident. Twins studies show a heritable component to the risk of developing persistent pain of up to 60%. Part of this risk is very likely to be epigenetic as well (see below). Polymorphisms of the genes related to catechol-O-methyltransferase (COMT) have been shown to influence pain sensitivity. Genetic testing may provide some insights into the management of post-injury pain.
Poor Sleep
In post-injury period it is important to get good sleep – poor sleep in a contributor to tramsitioning to chronic pain.
Psychological Variables
The importance of a patient’s psychological state prior to injury is a significant factor in predicting risk for post-injury pain. Predictors of transitioning to chronic post-injury pain include early life stress or trauma, depression, anxiety, posttraumatic stress, low self-efficacy, ineffective coping strategies and pain catastrophizing (the tendency to describe a pain experience in more exaggerated terms than the average person, to think about it more, and/or to feel more helpless about the experience).
These conditions contribute to both the magnification of acute pain as well as the chronification of pain. Additionally, when uncontrolled, they contribute to the risk of substance and opioid abuse as means of coping with pain. Therefore it is very important to aggressively treat these conditions to achieve the best pain outcomes. Certain medications effective for depression and anxiety may also have direct benefit for pain, particulaly nerve pain and are reviewed below. Non-pharmacologic anxiety-reducing strategies to promote peace of mind are also discussed below.
Pain catastrophizing is associated with greater levels of acute post-injury and chronic pain. Counseling regarding pain catastrophizing and expected outcomes can reduce the associated stress and may help prevent chronification of pain. Instruction for improving coping strategies and cognitive behavioral therapy to enhance self-efficacy may be effective in reducing reliance on analgesics and transitioning to chronic pain.
Understanding the Transition of Acute to Chronic Post-Injury Pain
A great deal of research is being perfomed in an effort to better understand more about how acute pain becomes chronic. Due to the overlapping goals of reducing both acute post-injury pain and the transitioning to chronic pain, the approach to achieving these goals are much the same. To best understand how to reduce the chronification of pain it is important to understand the neurophysiologic processes that contribute to it. These process include neuroinflammation, peripheral and central sensitization and modulation of descending pain pathways from the brain to the spinal cord.
Neuroinflammation
Recent research that looks at the transitioning of acute pain to chronic pain has identified neuroinflammation as a major contributor to the process. Neuroinflammation is an inflammatory condition within nerve tissue both peripherally and centrally, in the spinal cord and brain. It also contributes to post-injury depression, fatigue and cognitive dysfunction (impaired thought processing, or “brain fog”).
Inflammatory cells, called glial cells are found within the tissue matrix adjacent to and surrounding nerves, especially in the dorsal horn of the spinal cord. These glial cells respond to nerve injury as may occur with trauma and normally contribute to the healing process. Cells called mast cells play a role in the activation of glial cells. However, for reasons not entirely understood, the inflammatory response of glial cells may become poorly regulated leading to pathologic inflammation around the nerve which leads to chronification of pain that persists beyond the normal healing process.
A growing body of evidence suggests that regulation of mast cells and glial cells may facilitate the healing process and at the same time reduce chronic neuroinflammation thereby reducing the development of chronic pain. Unfortunately, at this point in time most of the research is limited to preclinical research (laboratory-based) and definitive answers to the prevention and treatment of chronic pain related to neuroinflammation is still in the early phases. However, some agents have been identified that research indicates are safe and effective in the management of glial cell regulation and may be effective for neuroinflammatory pain.
Treatments directed at neuroinflammation to reduce the transition of acute to chronic pain may also reduce the incidence and/or severity of post-injury depression, fatigue and cognitive dysfunction. At this time, the following agents are considered good candidates for treating neuroinflammation through stabilization and/or suppression of glial cells and mast cells:
- Palmitoylethanolamide (PEA)
- Cannabidiol (CBD)
- β-Caryophyllene
- Minocycline
- Low-dose naltrexone
See: Neuroinflammation
Peripheral and Central Sensitization
Chronic post-traumatic pain is generally considered to be largely neuropathic (nerve-related), sometimes occuring as a consequence of direct nerve injury such as transection, contusion, stretching, or inflammation of a nerve. Post-injury nerve pain also occurs as the result of sensitization of the peripheral and central nervous system as a consequence of continued pain.
Peripheral Sensitization
Following trauma, the intense influx of acute pain signals from peripheral nerves related to tissue trauma can trigger a magnification of pain through peripheral and central nerve sensitization, in which the excitability and responsiveness of nerves is increased. Sensitization in this circumstance refers to a condition of magnified sensory experience in which persisting pain becomes more severe over time as a result of continued pain signals transmitted by the peripheral nerves to the spinal cord and brain. This persisting pain signaling results in increased connections between nerves causing hyper-responsiveness to pain, including greater pain with lesser painful stimulation (hyperalgesia), pain with non-painful stimulation (allodynia), a magnified experience of pain and spread of pain sensitivity to non-injured areas.
Central Sensitization
After damage to the pain receptors (nociceptors ) from trauma, a hyperalgesic state can occur. This is divided into peripheral hyperalgesia, resulting from the sensitization of the peripheral nociceptors, and central hyperalgesia, which is associated with sensitization of the spinal cord and the central nervous system (central sensitization). Sensitization especially takes place in the dorsal horn of the spinal cord where peripheral nerves synapse (interact) with central nerves and pain transmission is enhanced. It is in this dorsal horn area that the impact of medications that modulate the descending pain pathways can occur to reduce pain perception and sensitivity. Contrary to common belief, peripheral and central sensitization can occur within a very short time span and can significantly contribute to a post-injury pain state.
Central sensitization is an important contributing process in the transition of acute to chronic pain. It is associated with many chronic post-traumatic pain syndromes, especially chronic neck and back pain. When treating acute pain it is important to take steps to avoid or suppress central sensitization however possible to prevent the pain from becoming chronic. These steps are reviewed below.
Prevention and treatment of peripheral and central sensitization remains somewhat elusive due to lack of a thorough understanding of the mechanisms involved in the sensitization process. What is known, however, is that a multi-modal approach seems to offer the most benefit and includes the use of medications, diet and nutrition, nutriceutical supplements and behavioral approaches.
For more information, see: Central Sensitization
Modulation of Descending Pathways
The dorsal horn of the spinal cord, where multiple pain mechanisms converge, serves as the interface between peripheral and central pain processing. This is where conscious and unconscious activity in the brain can influence the experience of pain severity and duration. It is also here that central sensitization, including hyperalgesia and allodynia appear to be initiated. A variety of mechanisms, pharmacologic and non-pharmacologic, exert their effects on the descending pain pathwys from the brain to the dorsal horn may reduce the transition of acute to chronic pain.
The pharmacologic methods include medications that act directly or indirectly on the descending pain pathways including duloxetine (Cymbalta), venlafaxine (Effexor), milnaciprin (Savella), the tricyclic antidepressants (TCAs) including amitriptyline (Elavil) and doxepin as well as certain opioids such as tramadol and tapentadol (Nucynta). Non-pharmacologic approaches include behavioral activities such as mindful exercises, meditation, prayer, listening to music and hypnosis (see below: Psychosocial Approaches to Reducing the Transition to Chronic Pain).
.
Treatment Interventions to Reduce the Transition of Acute to Chronic Post-Injury Pain
Treatment interventions to reduce the transition of acute to chronic pain are directed at reducing neuroinflammation and preventing or reducing peripheral and central sensitization.
Multi-modal Approach
Because severe, uncontrolled pre-injury and/or immediate post-injury pain significantly contributes to increased risk for the chronification of pain, it is important to treat pain aggressively in the initial post-injury period (see Acute Pain: Initial Post-Injury). When a patient with acute post-injury pain is identified as being at increased risk for transitioning from acute to chronic pain, however, additional treatment considerations are called for.
In an effort to reduce transitioning from acute to chronic pain, non-pharmacologic methods should include diet and nutrition-based nutriceuticals approaches, acupuncture, relaxation and music therapy and hypnosis as part of a multi-modal approach to reduce transitioning to chronic pain. Pharmacologic agents include neuromodulator medications, certain opioids, NMDA antagonists, muscle relaxers and cannabidiol (CBD).
The following is a review of recent research exploring ways to reduce the transition of acute to chronic post-injury pain.
Diet and Post-Injury Pain
Diet plays multiple roles in post-injury pain. To start with the obvious, it provides nutritional support for the healing process and as such should include nutrients favorable to repairing damaged tissues. It also plays a role in inflammation including, importantly, the resolution of the acute inflammatory process that is a normal and vital part of the healing process. Diet also plays a role in addressing oxidative stress which is a natural outcome of traumatic injury to tissues.
Omega-3 Fatty Acids
A significant contributing factor in the evolution of chronic pain is the failure of the acute pain and inflammation associated with a tissue injury to resolve as part of the healing process. Instead, when the inflammatory process fails to resolve, the chronification of pain ensues. Recent research has identified some of the variables in this process of inflammation resolution and the role post-trauma diet may have.
Preliminary research suggests that omega-3 fatty acids may play a role in resolving innappropriately persisting inflammation. As such, a diet rich in omega-3 may be helpful. Foods with the highest content of omega-3 include seafood and fish, especially mackerel, salmon, herring, oysters, sardines and anchovies. Other foods rich in omega-3 include flax seeds, chia seeds, walnuts, caviar and soy beans. Natural fish oil supplements also rich in omega-3 include salmon, sardines, herring and cod liver oil. These oils are available in capsules or liquid form.
See: Diet and Pain
Antioxidants
The oxidative stress associated with the traumatic destruction of tissue calls for a diet rich in antioxidants to aid in recovery and reduce inflammation. Some of the best food sources of antioxidants include dark chocolate or cacao, pecans, blueberries, strawberries, artichokes, goji berries, raspberries and kale. Antioxidant supplements that may offer benefit in reducing post-injury oxidative stress and inflammation. include vitamin C, curcumin and resveratrol.
See: Oxidative Stress
Anabolic Diet
An anabolic diet is one that supports healing processes involved in tissue repair and regeneration. Protein is an essential component in the healing process so it is recommended to maintain a diet high in protein to facilitate recovery after injury. Additionally, essential nutrients which are increased in demand by healing tissues including the B-vitamins and vitamin C. Minerals including calcium, magnesium and zinc are also important. Supplementing with a quality multi-vitamin, especially one with antioxidants such as PhytoMulti by Metagenics is advised. Better yet, make an appointment with our registered dietitian for a focused session on post-injury dietary considerations.
Nutriceutical Supplements
A nutriceutical is a pharmaceutical nutrient used for medicinal purposes. There are a multitude of supplements on the market with numerous health claims so it is important to identify those with good evidence for safety and therapeutic benefit. In the context of reducing or preventing chronification of pain, some nutriceuticals offer potential effectiveness in enhancing tissue healing, reducing pain and tissue inflammation, neuroinflammation and central sensitization.
The following nutriceuticals have evidence for safety and therapeutic benefit and offer potential usefulness in the setting of post-injury pain. Their proposed benefits are based largely on preclinical laboratory and animal research along with theoretical mechanistic activity.
Palmitoylethanolamide (PEA)
Perhaps the best agent currently recommended for neuroinflammatory pain is palmitoylethanolamide (PEA), a natural substance manufactured by glial cells and also found in foods such as milk and egg yolks. PEA has good evidence to support its benefit in regulating neuroinflammation through its stabilization of mast cells and glial cells. Another safe and potentially effective natural substance that may be effective in glial cell neuroinflammation is resveratrol.
See: Palmitoylethanolamide (PEA) & Resveratrol
Curcumin
Curcumin is a naturally occurring antioxidant and powerful anti-inflammatory with potency equivalent or superior to ibuprofen and other NSAIDs, but it is not associated with the toxicity and safety concerns associated with NSAIDs. Furthermore, it is a NRF2 activator and as such is believed to stimulate cellular production of endogenous antioxidants.
Additionally, curcumin has been found to be a histone deacetylase (HDAC) inhibitor. It is postulated that nerve injury may produce epigenetic changes such as histone deacetylation that may trigger the production of inflammatory mediators (See “Epigenetics” below). This is an epigenetic function that contributes to restoring an impaired noradrenergic descending inhibitory system by acting on noradrenergic nerves in the locus cereulus in the brain.These properties of curcumin all offers potential benefit in the setting of post-injury pain.
See: Curcumin and NRF2 activators
N-Acetyl Cysteine (NAC)
(More info coming)
Aromatherapy
Aromatherapy is an under-recognized and under-appreciated alternative form of treatment that consists of using aerosolized essential oils to achieve therapeutic benefit. Aromatherapy has shown benefit in reduction of pain and anxiety in the immediate post-injury period. Many of the aromatic compounds (terpenes) that have demonstrated therapeutic benefits with aromatherapy are also found in cannabis and contribute to the clinical benefits achieved with use of medical marijuana. For example, linalool found in lavender oil and cannabis, has proven effectiveness for sleep and anxiety.
Neuromodular Medications
There are a number of medications that offer potential benefit in reducing the chronification of post-injury pain although their use in this setting would be considered “off-label.” Medications directed at suppressing the development of neuroinflammation and central sensitization along with reducing neuropathic pain, include the gabapentenoid drugs [gabapentin (Neurontin) & pregabalin (Lyrica)], duloxetine (Cymbalta), venlafaxine ER (an extended release antidepressant), NMDA antagonists and glial cell inhibitors including PEA, minocycline, CBD and low-dose naltrexone.
Gabapentenoids: Gabapentin (Neurontin) & Pregabalin (Lyrica) in Acute Post-Injury Pain
Gabapentin (Neurontin)
Most studies evaluating the benefit of gabapentin in the transition of acute to chronic pain have been performed in post-operative conditions. In multiple reviews of studies evaluating post-operative pain after total knee and total hip arthroplasties (replacements), surgical procedures in which parts of the joints are replaced with artificial parts (prostheses), it was shown that the need for opioids for post-operative pain was reduced when gabapentin was used. Gabapentin trials show a moderate to large reduction in the occurrence of pain chronification altogh some recent studies has called this into question.
It should be noted that gabapentin is not without side effects that may include cognitive impairment, dizziness, memory impairment, nausea and diarrhea. It has also recently been demonstrated that gabapentin may also contribute to respiratory depression in combination with opiods. It is has not clear how frequent or clinically significant this respiratory depression is outside of an over-dose scenario, but it should be monitored and used with caution in those at high risk for central or obstructive sleep apnea.
Pregabalin (Lyrica)
Similarly, pregabalin (150 or 300 mg) has been studied pre-operatively and then continued postoperatively – in some studies, for only 2 doses, but in other studies, for up to 2 weeks. A systematic review and meta-analysis found that pregabalin significantly reduces the incidence of pain at 6 months and 12 months (4% vs 15%). Pregabalin does not seem to have the same contribution to respiratory depression as gabapentin.
Dosing
The optimal dosages of gabapentin and pregabalin for reducing the transition of acute to chronic post-injury pain are still controversial. Gabapentin doses may range from 300 mg to 1,200 mg/day, and those for pregabalin rmay anged from 50 mg to 300 mg/day for preventative purposes. When prescribed for acute symptomatic nerve pain, the maximum daily dose of gabapentin is 3600mg/day and for pregabalin 600mg/day.
Lidocaine
Lidocaine has been used for treatment of both acute and chronic pain for many years. It is commonly known for its use as a local anesthetic for numbing tissues prior to dental or minor surgical procedures such as suturing lacerations. However, it is not the numbing effect per se that provides lidocaine’s benefit for the management of pain. In fact, lidocaine has a number of different mechanisms by which it works for pain (see below). Evidence supports the use of lidocaine topically as well as by intravenous infusion for acute and chronic pain,
Topical Lidocaine
Lidocaine is used topically in many formulations, including ointments, creams and patches. Topical lidocaine is most commonly thought of as a treatment directed at acutely reducing pain, meaning providing relief in minutes or hours regardless of the underlying longevity of the pain condition be it acute or chronic.
The conditions for which topical lidocaine is most commonly used include nerve (neuropathic) pain such as post-herpetic neuralgia (shingles), peripheral neuropathy due to conditions including diabetes and post-radiation and post-chemotherapy and post-traumatic as well as painful scarring such as from burns or surgery. Evidence indicates that not only is the severity of pain reduced but also the size of the area of pain is lessened. Many patients also respond well when it is used for myofascial pain (muscle) which is also believed to have a neuropathic component. Additionally, it has been shown to decrease the area of hyperalgesia induced by topical capsaicin and sunburn.
Topical lidocaine to reduce transitioning of acute to chronic pain
Recent research has demonstrated that topically applied lidocaine reduces peripheral and central sensitization, conditions that contribute to the severity of chronic pain and the transition of acute to chronic pain. Due to the predeliction of shingles pain to transition to chronic persisting pain, the use of topical lidocaine early in the course of shingles as the open lesions resolve with the goal of not just managing immediate pain but also to reduce the transitioning to chronic pain. This research also strongly argues for the use of topical lidocaine in the setting of acute trauma, particularly post-surgical pain in conditions known to be most vulnerable for transitioning to chronic pain such as open chest surgery (thoracotomy), breast surgery, inguinal hernia surgery, and especially amputations.
Intravenous infusions of lidocaine
Lidocaine infusion is a safe, inexpensive and relatively easily administered treatment for neuropathic pain, typically dosed in a 2.5 – 5 mg/kg infusion protocol. Studies of lidocaine infusion in patients with diabetic neuropathy resulted in markedly reduced pain severity and quality at 14 and 28 days post infusion, highlighting its long-lasting effects.
Variables that contribute to the effectiveness of lidocaine infusion include both severity of pain and age of the patient. Each point increase of pain (in an 11-point scale) increased the odds of responding to lidocaine by approximately 29%, while each decade of life increased the odds by 36%.
Lidocaine infusion for fibromyalgia
Lidocaine infusion may also be beneficial in fibromyalgia. Studies evaluating serial infusions at intervals of 24 hours to 30 days demonstrated improvement that was sustained up to 3 to 5 weeks after the last infusion. Doses ranged from 1 mg/kg, up to 5 mg/kg of IV lidocaine, however 5 mg/kg was significantly more effective. The protocol in one center administers 5 mg of lidocaine per kg of body weight (maximum of 500 mg) over 1 hour diluted in 0.9% saline. Due to the antiarrhythmic property of lidocaine, patients undergo cardiac monitoring during the duration of the infusion and for 1 hour afterward. Vital signs are obtained every 10 minutes over the 2-hour period. Patients are premedicated with diazepam 10 mg (Valium) orally to reduce the theoretical risk of seizures from lidocaine in the central nervous system, although no seizures were noted. With regard to the interval schedule, our patients who benefit from lido‐ caine infusions appear to have duration of benefit of 3 to 5 weeks and are scheduled accordingly.
Lidocaine infusion for neuropathic back pain
One 2012 study investigated the effects of intravenous lidocaine on neuropathic pain in failed back surgery syndrome (FBSS) patients. In this study, both 1 mg/kg, and 5 mg/kg of IV lidocaine,improved pain but the 5 mg/kg dose was significantly more effective. Another study evaluated its use for acute low back pain with sciatica but no benefit was found.
Mechanisms of Action of Lidocaine
Lidocaine has multiple modes of actions that explain its clinical role in treating peripheral and central pain. It has potent anti-inflammatory properties that are more potent than traditional anti-inflammatory drugs, with fewer side effects. Lidocaine infusion has been shown to reduce circulating inflammatory cytokines that contribute to hyperalgesia and central sensitization, suggesting a mechanism for its effectiveness in reducing the mechanical allodynia and hyperalgesia associated with chronic neuropathic pain.
On a cellular level, lidocaine blocks sodium channels in nerves, uncouples G protein and has been reported to also block NMDA receptors.
Mechanisms of Action of Lidocaine in Peripheral and Central Sensitization
Pain conditions often begin with damage to peripheral tissues and/or nerves, leading to enhanced transmitter release and nerve conduction to the spinal cord. When this persists, these hyperexcitable nerves become sensitized and show reduced thresholds, greater evoked responses, increased receptive field sizes, and ongoing stimulus-independent activity that lead to the allodynia, hyperalgesia, and spontaneous pain that characterize central sensitization..
By blocking the sodium channels of hyperexcited, repetitively activated primary afferent nerves (notably C fibers and Aδ fibers), lidocaine suppresses the peripheral sensitization that drives central sensitization. Mounting evidence indicates that treatment strategies for peripheral and central sensitization require a multimodal approach. For example, a synergistic benefit may be obtained through the concomitant use of gabapentin (Neurontin) or pregabalin (Lyrica) with lidocaine based on their mechanism of blocking calcium channels of hyperexcited nerves.
Antidepressants
It is believed that the transition of acute to chronic pain involves nerve pathways that descend from pain centers in the brain (locus coeruleus) to the spinal cord (dorsal horn). These pathways may suppress or enhance pain signals coming from the spinal cord to the brain and thus may impact the perception of pain. These descending pathways involve two neurotransmitters, serotonin and noradrenaline, the neurotransmitters modulated by antidepressants and certain other medications including clonidine and tapentadol (Nucynta).
The mechanisms by which these medications modulate the descending pathways to reduce pain involve both suppressing nerve activity as well as reducing neuroinflammation by their action on glial cells in the spinal cord. Reduced noradrenaline activity in these areas is believed to contribute to the development of chronic pain and medications that enhance noradrenaline are helpful for reducing pain.
Antidepressants that reduce pain by enhancing noradrenaline activity include the serotonin and noradrenaline reuptake inhibitors [SNRIs, including duloxetine (Cymbalta), venlafaxine (Effexor) and milnacaprin (Savella)] and the noradrenaline reuptake inhibitors [NRIs, including the tricyclic antidepressants (TCAs), amitriptyline (Elavil) and doxepin]. Use of these medications has been shown to reduce neuropathic pain and may reduce the chronification of post-injury pain. However, research identifying their definitive benefit for the transition of acute to chronic pain remains inadequate. Therefore their use in post-injury pain is directed at acute symptomatic relief while offering potential prophylaxis against chronification of acute to chronic post-injury pain.
Alpha-2 Adrenergic Agonists
Noradrenaline, an important neurotransmitter in the pain centers of the brain and descending pain pathways from the brain to the spinal cord, has specific action mainly on adrenergic receptors. As noted above, reduced noradrenaline activity in these areas contributes to the development of chronic pain. Both α2- and β2-adrenergic receptors are present in microglia, and their activation leads to reduced neuroinflammation triggered by microglia. Medications that enhance noradrenergic activity also exert an anti-inflammatory effect, ultimately leading to analgesia.
The α2-adrenergic agonist medications (drugs that stimulate adrenaline and noradrenaline receptors) are a class of drugs used to treat multiple, distinctively different, conditions including high blood pressure, anxiety, opioid and other drug withdrawal and, importantly, pain. Common α2-adrenergic receptor agonists include clonidine (Catapres) and lofexidine (Lucemyra) and also tizanidine (Zanaflex), a medication commonly used as a muscle relaxer.
Clonidine (Catapres)
Systemic clonidine used in the peri-operative period has been shown to decrease pain intensity and nausea post-operatively for up to 24 hours or longer as well as having a sparing effect for the use of opioids for acute post-operative pain. In other studies, oral clonidine doses between 0.1 to 0.3 mg have been used as a pre-medication to reduce post-surgical pain. Oral clonidine has been shown to reduce anxiety and pain for up to 72 hours after surgery. Unfortunately at this time there are no studies evaluating the benefit of clonidine for prevention of chronic post-surgical or post-injury pain.
See Alpha-2 Adrenergic Agonists
Tizanidine (Zanaflex)
Tizanidine is frequently used to treat myofascial pain in the head and neck. It reduces painful muscle spasms in the neck and shoulder and can reduce spasticity by increasing the presynaptic inhibition of motor neurons in the brain and spinal cord. Tizanidine also has α2-adrenergic agonist activity that may contribute to additional benefit for reducing neuroinflammation.
NMDA Antagonists
An important mechanism in hyperalgesia involves the N-methyl-D-aspartate (NMDA) receptor, known for its major role in nerve plasticity (modification), and glutamate, the neurotransmitter that activates it. NMDA activity has been shown to contribute to central sensitivity and the development of chronic pain, suggesting a role for the use of NMDA antagonists for potentially reducing both immediate post-injury pain as well as for reducing the risk of the chronification of post-injury pain.
While the clinical data on the use of NMDA antagonists is growing, no definitive regimens have been defined for their use in the post-injury setting despite increasing preclinical evidence for their benefits. So far, the only studies apparently available for the use of NMDA antagonists in the peri-operative setting focus on ketamine (see below). In principle, however, other medications with excellent safety profiles and NMDA antagonist activity are available, prescription and non-prescription.
While the NMDA activity of some of these medications may be low, it is often the case in the field of medicine that the amount of a medication used to prevent a condition is less than that required to reverse a condition. While studies are lacking for efficacy, generally speaking the safety of these medications is well established.
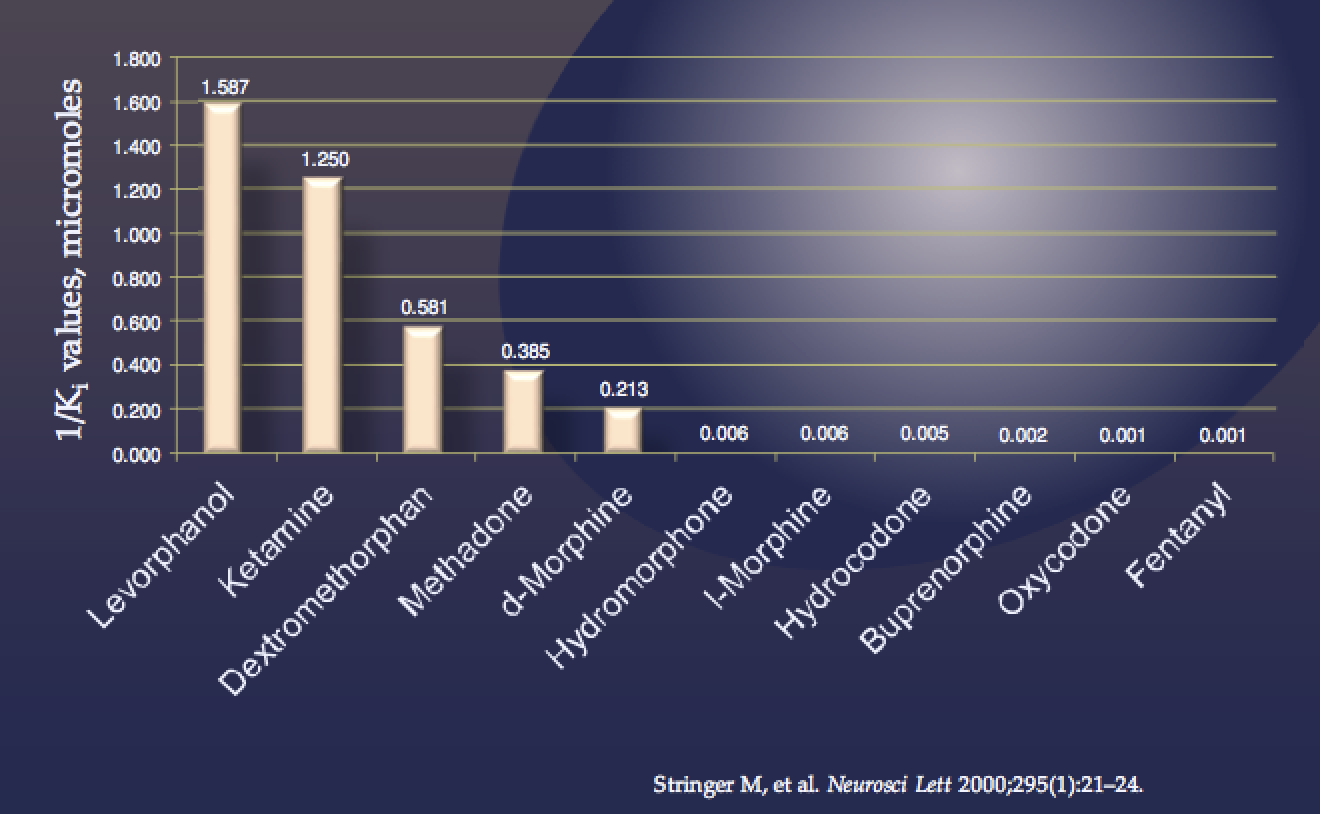
Different classes of drugs demonstrate NMDA antagonism to a range of degrees. Perhaps the most useful drugs with this benefit are the opioids and specifically levorphanol for use in acute post-injury pain.
NMDA antagonist activity of various opioids
Choice of Opioids for Post-Injury Pain
Opioids remain the analgesics of choice for post-injury pain control for moderate to severe pain. Since the severity and duration of post-injury pain is a risk factor for transitioning of acute to chronic pain, opioids can help in preventing this transition. While strong opioids may be associated with opioid-induced hyperalgesia, likely transient, good acute pain control is important for preventing pain chronification and aggressive control of severe pain should be emphasizeed. Interestingly, patients with negative beliefs about opioids have been shown to have a higher risk for transitioning of acute to chronic pain.
Due to current concerns regarding the “opioid epidemic” and recent research evaluating the tendency for opioids taken for acute pain to become chronically prescribed, the use of opioids for acute pain needs to be directed carefully by the prescribing physician. It is strongly recommended that patients should use the lowest effective dose of opioids for the shortest period possible. Additionally, the use of extended release opioids are generally avoided in the acute management of pain, in part because they may have a higher rate of abuse and diversion, and because they may contribute to the chronification of opioid use.
For more information, see: Transition of Acute Post-Injury Opioid Use to Chronic Opioid Use.
When opioids are necessary for the management of acute post-injury pain, it is important to consider which choices of opioids may offer the best likelihood in reducing the transition of acute to chronic pain. While research is lacking in this area, there are principles surrounding the mechanisms of acute pain transitioning to chronic pain and the mechanisms of action of different opioids that suggest some opioids may be superior to others under these circumstances and they are summarized here.
Tramadol (Ultram)
Tramadol is frequently the first opioid prescribed for mild to moderate pain and it has the advantage of offering potentially more benefit for nerve pain compared with many other opioids due to its secondary activity on the descending pain pathways. It has the disadvantage of being subject to genetic variants in which a patient may be unlikely to experience analgesic benefit (7% of Caucasians, about 1% of Orientals) or may have enhanced benefit with the potential for more side effects (about 2% of people).
Buprenorphine
Buprenorphine is an opioid that is considered to be a partial opiate agonist which imparts special properties not shared by traditional opioids. It has the advantage of providing up to 12 hours of analgesic benefit per dose in the case of Belbuca, a film strip that is applied to the inside cheek. In the case of Butrans, buprenorphine is formulated as a topical skin patch that can be worn for 7 days while providing consistent analgesic benefit 24 hours/day.
Additionally, buprenorphine offers greater benefit for nerve pain and it develops tolerance very slowly compared with most traditional opioids. Most importantly, it imparts no significant respiratory depression as a side effect which makes buprenorphine the safest opioid for avoiding accidental overdose. Buprenorphine is a preferred opioid for patients who are elderly or for those with sleep apnea or prescribed other sedating medications.
Some physicians believe that because of buprenorphine’s special properties it should be preferentially considered for early or initial use in acute pain if initial management with usual short-acting opioids is ineffective, not tolerated or less safe compared with buprenorphine.
Levorphanol
Levorphanol is an opioid with special characteristics including potent NMDA antagonism that makes it a particularly attractive choice for acute post-injury pain management, especially in those at risk for transitioning to chronic pain. While appropriate for use on an as-needed basis, it offers a longer half-life compared with usual short-acting opioids including hydrocodone, oxycodone and hydromorphone.
See: Levorphanol
Tapentadol (Nucynta)
Tapentadol has proven benefit in the management of both acute and chronic pain and is effective in treating acute post-operative pain. It may, however, have additional advantage for reducing chronic postsurgical pain (CPSP) because of its action on the descending pain pathways, noradrenaline transmission and alpha-2 adrenoceptors activation which has been shown to reduce neuroinflammation.
A recent study found that a single preemptive oral dose of tapentadol (75 mg) is effective in reducing perioperative analgesic requirements and acute postoperative pain, without added side effects. Additionally, another study has demonstrated a synergistic effect with combining palmitoylethanolamide (PEA) with tapentadol in reducing chronic back pain.
Adjunctive NMDA Antagonists for Pain
NMDA antagonists, especially ketamine and magnesium, have been shown to reduce post-operative pain but there are few studies related to reducing post-injury pain. Ketamine has a clear opioid-sparing effect in the perioperative period and may even reduce long-term opioid consumption in opioid-tolerant patients. Unfortunately, ketamine has been studied almost exclusively only for intravenous use. Magnesium has produced mixed results as a postoperative analgesic when used alone but has demonstrated synergism when combined with morphine or ketamine (See below).
Ketamine
Animal studies show that medications that block NMDA can abolish hyperalgesia, supporting the rationale of using ketamine in the management of post-operative pain. A large study published in 2015 reviewing post-operative pain in adults confirmed the benefits of both ketamine and magnesium in reducing post-operative pain intensity and improved patient satisfaction. The benefits of the perioperative use of a subanesthetic dose of ketamine for the prevention of various types of CPSP, including phantom limb pain, has been supported by multiple studies. Significant reduction in pain for up to 6 months postoperatively has been seen with intravenous ketamine in patients undergoing colon resection.
Unfortunately, the use of ketamine intravenously (IV) does not lend itself to usual acute post-injury pain management. This is largely because the side effects of IV ketamine can be significant, including an acute dissociative psychologic effect, though rare. It is currently recommended that IV ketamine use be limited to special clinical situations.
Intranasal Ketamine
The use of ketamine as a nasal spray has receive attention recently and appears to be safe and effective in studies evaluating its use for acute pain, including post-injury pain in children and adults. While onset of pain relief occurs rapidly within 15 minutes, peaks in effectiveness at about 60 minuts and lasts for a few hours. Ketamine IV is being studied intensively for treatment of severe depression with excellent results but studies assessing ketamine for acute pain management remains limited, especially in the United States.
Topical Ketamine
Ketamine can also be applied topically which has been shown to be effective for nerve pain, especially in peripheral neuropathies and shingles. It offers potential benefit for peripheral neuropathy but has not been studied. Topical ketamine is only available by prescription as compounded formulation and may only be filled by a compounding pharmacy.
See Ketamine
Topical Lidocaine
Lidocaine is an anesthetic numbing agent that is available in prescription 5% patches that can be applied to a painful area for up to 12 hours/day. The lidocaine has a direct action on the sodium channels on nerves, it is not the numbing effect of lidocaine that contributes tio its pain benefit (see above). Over the counter lidocaine creams are also available from most pharmacies.
Dextromethophan (DM)
Dextromethorphan (DM) is a common ingredient in cough and cold remedies, including Delsym, Robitussin DM and many others. DM is structurally related to the opioid, levorphanol, although DM is not itself considered an opioid. As an NMDA antagonist, dextromethorphan when used as an adjunctive to opioids for perioperative pain may be useful in reducing pain, especially neuropathic pain, as well as preventing nerve damage and hyperalgesia. While research remains conflicting and still falls short of confirming definitive benefits and doses for supplementing with DM, there are evidence-based arguments to consider including a trial use of dextromethorphan in the management of post-injury pain.
Orphenadrine (Norflex), a medication used for muscle spasm, also has weak NMDA activity and may offer benefit in reducing the development of hyperalgesia. When there are indications for the use of a muscle relaxer in the post-injury period, orphenadrine may offer an advantage.
Memantine (Namenda)
Memantine (Namenda), a medication used for Alzheimer dementia, has NMDA activity and there is growing evidence suggesting that memantine may be useful in reducing neuropathic (nerve) pain. Memantine has been studied in certain subgroups of pain patients including a trial showing the benefit of a combination of morphine and memantine in patients with complex regional pain syndrome (CRPS).
Magnesium Sulfate (MgSO4)
Magnesium given intravenously, has been shown in multiple recent studies to reduce post-operative pain and allow for reduced dosing of opioids. In a March 2017 study, investigators concluded that MgSO4 appeared to increase the analgesic potency of morphine. While the mechanism of this action has not been clearly defined, it is consistent with NMDA antagonist activity. A 2013 meta-analysis concluded that peri-operative intravenous magnesium reduced opioid needs and pain scores in the first 24 hours postoperatively, without any reported serious adverse effects.
While it is not generally recommended to give magnesium intravenously for acute post-injury pain, the use of topical magnesium through the use of topical magnesium creams or epsom baths (magnesium sulfate) allows for topical absorption through the skin and may offer benefits for reducing the transition to chronic pain.
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs)
As noted above, peripheral sensitization of nociceptors leading to primary hyperalgesia is an important contributor to post-injury pain. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs), which reduce peripheral prostaglandin concentration and lead to reduced peripheral sensitization are a useful component of multimodal analgesia. In randomized controlled trials and their meta-analyses, NSAIDs improve analgesia, reduce opioid requirements, and reduce adverse effects of opioids, including reducing postoperative nausea and vomiting associated with opioids in the early postoperative period.
However, despitee their established benefit for reducing acute pain, there is no evidence available that NSAIDs will reduce peripheral or central sensitization or reduce the transition from acute to chronic pain. In fact, there is early, preclinical evidence that NSAIDs may actually contribute to inappropriate prolongation of neuroinflammation and , in turn, contribute to peripheral and central sensitization. The resolution of neuroinflammation has previously been considered a passive process. Recent research, however, has identified endogenous mediators that actively resolve inflammation called resolvins, protectins & maresins. Production of these agents may be promoted by low dose aspirin and omega-3 essential fatty acids while NSAIDs may inhibit their production and contribute to inappropriate prolongation of neuroinflammation.
See: Neuroinflammation
Caution should be exercised with use of all NSAIDs in patients with kidney disease and those with increased risk of heart disease or stroke. One of the most important concerns with use of NSAIDs is the increased risk of postoperative bleeding with NSAIDs although a meta-analysis has shown that ketorolac (Toradol) does not increase the risk of perioperative bleeding. Additionally, celecoxib (Celebrex), a COX-2 selective NSAID, provides similar effectiveness to nonselective NSAIDs, but is superior in the postoperative setting because of its reduced incidence of gastritis and gastrointestinal bleeding compared with nonselective NSAIDs.
Celecoxib lacks platelet inhibitory effects and therefore causes less postoperative blood loss than nonselective NSAIDs, comparable to placebo. Additionally, Celecoxib shows a rate of gastric ulceration similar to placebo and significantly lower than nonselective NSAIDs in high-risk patients, even for short-term use. Celecoxib does not cause bronchospasm in patients with NSAID-exacerbated respiratory disease. Concerns about cardiovascular complications of COX-2 selective NSAIDs has not been demonstrated with Celecoxib with either short-term or long-term use.
One note regarding celecoxib:
Concerns have been raised that there may be cross-reactivity between allergies to sulfa antibiotics and celecoxib, due to a structural sulfonamide element of the celecoxib shared with sulfa antibiotics. Because of this, it is often advised to avoid celecoxib if one has an allergy to sulfa antibiotics because a history of sulfa allergy may be associated with an increased risk of adverse reactions to a wide range of nonantibacterial sulfonamides, including celecoxib, loop and thiazide diuretics, certain antivirals, carbonic anhydrase inhibitors, and sulfonylureas. Concerns have also been raised that patients who have experienced an allergic reaction to one nonantibacterial sulfonamide may be at risk for an adverse reaction to others. However, there is good evidence that none of the nonantibiotic sulfonamides exhibit both of the features shown to be responsible for sulfonamide allergic reactions. The weight of evidence suggests that withholding nonantibacterial sulfonamides such as celecoxib from patients with prior reactions to antibacterial sulfonamides or other nonantibacterial sulfonamides is not clinically justified.
See: NSAIDs
Topical Medications
The use of topical medication are recommended for the management of acute pain as well as the reduction of neuroinflammation and peripheral and central centralization. A study of topical 1% amitriptyline, 0.5% ketamine or their combination for the treatment of neuropathic pain found the combination cream to be effective after 7 days’ application, and more effective than when either were applied individually.
Topical Ketamine
Ketamine can also be applied topically which has been shown to be effective for nerve pain, especially in peripheral neuropathies and shingles. It offers potential benefit for peripheral neuropathy but has not been studied. Topical ketamine is only available by prescription as compounded formulation and may only be filled by a compounding pharmacy.
See Ketamine
Topical Lidocaine
Lidocaine is an anesthetic numbing agent that is available in prescription 5% patches that can be applied to a painful area for up to 12 hours/day. The lidocaine has a direct action on the sodium channels on nerves, it is not the numbing effect of lidocaine that contributes tio its pain benefit. Over the counter lidocaine creams are also available from most pharmacies.
Topical Magnesium
The use of topical magnesium through the use of topical magnesium creams or epsom baths (magnesium sulfate) allows for topical absorption through the skin and may offer benefits for reducing the transition to chronic pain based on its action as an NMDA antagonist. Magnesium offers other benefits for muscle pain as well.
See: Magnesium
Glial cell inhibitors
Glial cells, the most common of which are microglia cells and astrocytes, are cells found in the central and peripheral nervous system. Glial cells provide a supportive matrix for nerve cells, supplying nutrients and oxygen and aid in the repair of damaged cells. They function to maintain balance in nerve and neurotransmitter activity and provide support and protection for neurons (nerve cells). However, when activated, glial cells also are important in the evolution and maintenance of chronic nerve pain through the release of pro-inflammatory peptides that trigger neuroinflammation and plays a significant role in the evolution of central sensitization.
Research is still early regarding the intervention on glial cell activity to prevent neuroinflammation and peripheral and cental sensitization. However, there is evidence that some medications and nutriceuticals may be of benefit, including minocycline, palmitoylethanolamide (PEA) and low dose naltrexone.
Minocyline
Minocycline (Minocin) is an antibiotic sometimes used to treat rheumatoid arthritis (RA). It is part of the group of antibiotics known as tetracyclines. Although RA is not thought to be caused by an infection, minocycline may improve the signs and symptoms of this disease. Aside from its antibiotic property, minocycline has been show in inhibit the activation of glial cells and consequently reduce the process of neuroinflammation. For this reason minocycline has been proposed to offer benefit in the post-operative period to reduce the development of central sensitization and chronic pain. Studies investigating this proposed benefit are as yet inconclusive however and it may be premature to suggest minocycline as a means of reducing the transition of acute to chronic post-injury pain at this time.
See Minocycline
Low-dose Naltrexone (LDN)
Low-dose naltrexone (LDN) refers to a prescription drug that acts as an opioid antagonist but is used at very low doses that would offer minimal effectiveness for its usual application at usual doses. LDN is used at doses of 4.5 mg to 6 mg instead of usual doses of 100 mg naltrexone which is prescribed to help prevent relapses in alcohol or drug abuse. LDN is throught to suppress microglia cells and reduce central sensitivity. It has been proposed as effective in a number of chronic pain conditions associated with central sensitivity (e.g. fibromyalgia, complex regional pain syndrome, migraine headache, interstitial cystitis). It has not been studied in reducing the transition of acute to chronic pain but might theoretically provide benefit. Because it blocks opioids it is not recommended to use along with opioids.
See:
(3) Palmitoylethanolamide (PEA)
Psychosocial Approaches to Reducing the Transition to Chronic Pain
Psychosocial approaches to reduce pain, limit psychological distress and improve comfort and self-efficacy are encouraged. The goals of these interventions are to improve overall mental health and decrease rates and severity of depression, anxiety, and posttraumatic stress disorder. Interventions include mindfulness interventions, music therapy, cognitive behavior therapy, hypnosis, self- management interventions and training, educational information access, peer support, and online social networking.
Mindfulness
Mindfulness trains an individual to observe painful situations as they arise and to consciously let go of the anxiety and stress associated with the painful event. The effectiveness of mindfulness training is best supported by studies of chronic pain but several studies have also shown that in the acute setting of pain mindfulness interventions can reduce perceived pain, pain intensity, and complicating factors (e.g., anxiety, anger, and depression). It is believed that mindfulness exercises reduce the central sensitization process that contributes to the transitioning to chronic pain.
Music Therapy
Music therapy is a passive technique that is self-explanatory, low-risk, and requires no caregiver expertise. Studies of hospitalized patients with acute pain demonstrate reductions in pain perception and improved mood scores after listening to music of their choice. Engaging the use of music decreases anxiety and improves sleep in the setting of pain while also allowing for reduced use of opioids. A meta-analysis of studies evaluating the specific characteristics of music used in reducing pain identified the most benefit for music without lyrics.
Cognitive Behavioral Therapy (CBT)
CBT focuses on improving physical and emotional functioning despite the pain, rather than attempting to eliminate the pain. It provides positive reinforcement to wellness behavior, physical fitness and cognitive reframing so that patients can become desensitized to the persistent pain and can function better. It usually combines stress management, problem solving, goal setting, relaxation, and the pacing of activities.
Cognitive behavioral interventions (CBT) can improve a patient’s control over their pain perception and provide pain relief. Examples of CBT include setting expectations before surgery, teaching relaxation techniques and using guided mental imagery for isolating pain perception. CBT can reduce symptoms in post-traumatic stress disorder (PTSD) trauma victims. There is evidence that web-based cognitive behavioral therapy is effective. Aromatherapy has been shown to improve anxiety tand pain.
Hypnosis
Hypnosis is a cognitive technique designed to heighten an individual’s responsiveness to suggestion in an effort to alter behavior, feelings, thoughts or perceptions. Hypnosis is used extensively for chronic pain syndromes but evidence also supports its use in acute injuries with a beneficial effect on pain and emotional distress for some patients.
Epigenetics
One of the worst outcomes of acute, post-injury pain is having that pain evolve into chronic pain. Understanding the mechanisms of how acute pain becomes chronic rather than healing and resolving is an important riddle that eludes full understanding.
Under persistent pain conditions cells processing pain signaling, i.e., nociceptors (pain receptors) in the peripheral nervous system and neurons/glia in the central nervous system, become sensitized in response to various stimuli. This increased sensitivity is accompanied by functional and structural changes (plasticity). Multiple mechanisms are likely responsible for these changes.
One area of study that is beginning to shed light on this process is the study of “epigenetics,” which refers to changes in a chromosome (DNA) that affect gene activity and expression. People inherit genes that determine personal characteristics including such as eye color and height or physiologic processes such as how medications are metabolized or how effective they are. The expression of these genes can be modified, for example by being amplified, suppressed or turned on or off. It is believed that epigenetic modification of genes may at least be in part responsible for transitioning acute pain, such as post-injury pain, to becoming chronic and persisting beyond apparent tissue healing. Understanding this process may lead to avoiding the evolution of chronic pain.
Medications can impact epigenetic change, including opioids. Both the developmental expression of the μ opioid receptor, and pathological hyperalgesia are mediated, at least in part, by epigenetic mechanisms. In general, opiates seem to increase global DNA methylation levels, a mechanism associated with hyperalgesia. This is in contrast to another group of drugs used in surgery – local anaesthetics. Local anaesthetics such as lidocaine appear to induce DNA demethylation, and may thereby protect against the development of hyperalgesia. Clinically, some studies have demonstrated a reduction of hyperalgesia and inflammation post-operatively via epigenetic mechanisms with local anaesthetics.
Additionally, curcumin has been found to be a histone deacetylase (HDAC) inhibitor. It is postulated that nerve injury may produce epigenetic changes such as histone deacetylation that may trigger the production of inflammatory mediators.
Of major significance is that epigenetic modification can be inherited, passed on to the next and future generations. In other words, an environmentally-induced change in DNA expression can affect not just the person but their children and grandchildren. In animal studies of stress-induced intestinal pain hypersensitivity, epigenetic changes associated with experimental irritable bowel disease could still be detected in offspring two generations later. It is believed also that during pregnancy exposure to certain medications or drugs can precipitate epigenetic changes that are then passed on to the fetus.
The study of epigenetics will most certainly have an important impact on the field of medicine in the decades to come.
One underlying principle should be emphasized:
Pain is magnified by stress. There are many measures one can embrace to reduce stress during the post-injury time frame. These measures include nutritional and behavioral options. It is strongly advised to explore these areas on other pages of this web site:
See:
References:
Preventing Transition of Acute to Chronic Pain – Overviews
- Preventing chronic pain following acute pain – risk factors, preventive strategies, and their efficacy – 2011
- Transition from Acute to Chronic Pain – 2008
- Transition from acute to chronic pain – 2015
- The Transition of Acute Postoperative Pain to Chronic Pain An Integrative Overview of Research on Mechanisms – PubMed – 2017
- The Emotional Brain as a Predictor and Amplifier of Chronic Pain – 2016
- Chronic postsurgical pain – current evidence for prevention and management – 2018
- The efficacy and safety of first-line therapies for preventing chronic post-surgical pain – a network meta-analysis – 2018
- A systematic review of therapeutic interventions to reduce acute and chronic post-surgical pain after amputation, thoracotomy or mastectomy – 2014
- The_Transition_from_Acute_to_Chronic_Pain_Might_In Risk Factors Associated With Transition From Acute to Chronic Low Back Pain in US Patients Seeking Primary Care – 2021
- The transition from acute to chronic pain – might intensive care unit patients be at risk? – 2012
- Transitioning from Acute to Chronic Pain – An Examination of Different Trajectories of Low-Back Pain – 2018
- Preventing chronic pain following acute pain – risk factors, preventive strategies, and their efficacy – 2011
- Strategies to Treat Chronic Pain and Strengthen Impaired Descending Noradrenergic Inhibitory System – 2019
- Pathophysiology of musculoskeletal pain – a narrative review – 2021
- chronic post-hernia repair pain-2023
Oxidative Stress – Pain
- Roles of Reactive Oxygen and Nitrogen Species in Pain – 2011
- Clinical Relevance of Biomarkers of Oxidative Stress – 2015
- The Interplay between Oxidative Stress, Exercise, and Pain in Health and Disease – Potential Role of Autonomic Regulation and Epigenetic Mechanisms – 2020
- Neuropathic Pain – Delving into the Oxidative Origin and the Possible Implication of Transient Receptor Potential Channels – 2018
Ultrasound Therapy
Myofascial Pain
- Effectiveness of Ultrasound Therapy on Myofascial Pain Syndrome of the Upper Trapezius – Randomized, Single-Blind, Placebo-Controlled Study – 2018
- Myofascial Pain Syndrome in the Elderly and Self-Exercise – A Single-Blind, Randomized, Controlled Trial – 2016
- Effect of Therapeutic Sequence of Hot Pack and Ultrasound on Physiological Response Over Trigger Point of Upper Trapezius – 2015
- Effectiveness of ultrasound therapy for myofascial pain syndrome – a systematic review and meta-analysis – 2017
- Randomized controlled study of the antinociceptive effect of ultrasound on trigger point sensitivity novel applications in myofascial therapy? – PubMed – 2007
Preventing Pain Related to Surgery – Overviews
- perioperative-pain-management- 2007 pubmed-ncbi
- Pharmacotherapy for the prevention of chronic pain after surgery in adults (Review) – 2013
- optimizing-pain-management-to-facilitate-enhanced-recovery-after-surgery-pathways 2015 -pubmed-ncbi
- development-of-a-management-algorithm-for-post-operative-pain-mapp-after-total-knee-and-total-hip-replacement-2014
- Post-operative Opioid-Induced Hyperalgesia – ICM Case Summaries – 2016
- The Complexity Model – A Novel Approach to Improve Chronic Pain Care – 2014
- Postoperative_pain_from_mechanisms_to_treatment.
- Mechanisms of acute and chronic pain after surgery: update from findings in experimental animal models. – PubMed – NCBI – 2018
- Short-term pre- and post-operative stress prolongs incision-induced pain hypersensitivity without changing basal pain perception – 2015
- Age and preoperative pain are major confounders for sex differences in postoperative pain outcome – A prospective database analysis – 2017
- Optimizing pain management to facilitate Enhanced Recovery After Surgery pathways. – PubMed – NCBI – 2015
- Reward Circuitry Plasticity in Pain Perception and Modulation – 2017
- Surgically-Induced Neuropathic Pain (SNPP) – Understanding the Perioperative Process – 2013
- Preventing Chronic Pain following Acute Pain – Risk Factors, Preventive Strategies, and their Efficacy – 2011
- Regular physical activity prevents development of chronic pain and activation of central neurons – 2013
- Regional anaesthesia to prevent chronic pain after surgery – a Cochrane systematic review and meta-analysis – 2013
- Pharmacotherapy for the prevention of chronic pain after surgery in adults – 2017
- A systematic review of therapeutic interventions to reduce acute and chronic post-surgical pain after amputation, thoracotomy or mastectomy. – 2015
- Analysis of perioperative pain management in vascular surgery indicates that practice does not adhere with guidelines – a retrospective cross-sectional study – 2017
- Efficacy and safety of multimodal analgesic techniques for preventing chronic postsurgery pain under different surgical categories – a meta-analysis – 2017
- Postoperative pain—from mechanisms to treatment – 2017
- Local anaesthetics and regional anaesthesia versus conventional analgesia for preventing persistent postoperative pain in adults and children – 2018
- Chronic postsurgical pain – current evidence for prevention and management – 2018
- Preventive analgesia and novel strategies for the prevention of chronic post-surgical pain. – (2015)
- Poorly controlled postoperative pain – prevalence, consequences, and prevention – 2017
- Designing the ideal perioperative pain management plan starts with multimodal analgesia – 2018
- Special indications for Opioid Free Anaesthesia and Analgesia, patient and procedure related: Including obesity, sleep apnoea, chronic obstructive … – PubMed – NCBI – 2017
- Chronic pain patient and anaesthesia – 2019
- New Persistent Opioid Use After Minor and Major Surgical Procedures in US Adults – 2017
- Multimodal Analgesia, Current Concepts, and Acute Pain Considerations. – PubMed – NCBI -2017
- Recent Advances in Postoperative Pain Management – 2010
- Effect of perioperative systemic alpha2-agonists on postoperative morphine consumption and pain intensity – systematic review of randomized controlled trials- 2011
- Cannabinoids for Postoperative Pain – 2007
- Educating Patients Regarding Pain Management and Safe Opioid Use After Surgery – 2020
Preventing Pain Related to Surgery – Phantom Limb Pain
- Strategies for prevention of lower limb post-amputation pain: A clinical narrative review – 2018
- Optimized perioperative analgesia reduces chronic phantom limb pain intensity, prevalence, and frequency: a prospective, randomized, clinical trial. – PubMed – NCBI – 2011
- Chronic post-amputation pain – peri-operative management – Review 0- 2017
Preventing Pain Related to Surgery – Enhanced Recovery After Surgery (ERAS)
- Enhanced Recovery After Surgery: A Review. – PubMed – NCBI – 2017
- Pain management within an enhanced recovery program after thoracic surgery – 2018
- Optimizing pain management to facilitate Enhanced Recovery After Surgery pathways. – PubMed – NCBI – 2015
Preventing Pain Related to Surgery – Neuroinflammation, Glial Cells & Mast Cells
- Involvement of mast cells in a mouse model of postoperative pain. – PubMed – NCBI – 2011
- An Inflammation-Centric View of Neurological Disease – Beyond the Neuron – 2018
- Rescue of Noradrenergic System as a Novel Pharmacological Strategy in the Treatment of Chronic Pain – Focus on Microglia Activation – 2019
Preventing Pain Related to Surgery – Resolving Inflammation
- Vagus nerve controls resolution and pro-resolving mediators of inflammation – 2014
- The Resolution Code of Acute Inflammation – Novel Pro-Resolving Lipid Mediators in Resolution – 2015
- Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. – PubMed – NCBI – 2018
- Resolvins and protectins – mediating solutions to inflammation – 2009
- Resolvins and inflammatory pain – 2011
- Resolution of inflammation – an integrated view – 2013
- Protectins and maresins – New pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome – 2014
- Proresolving lipid mediators and mechanisms in the resolution of acute inflammation – 2014
- Novel Pro-Resolving Lipid Mediators in Inflammation Are Leads for Resolution Physiology – 2014
- Novel Anti-Inflammatory — Pro-Resolving Mediators and Their Receptors – 2011
- Lipid Mediators in the Resolution of Inflammation – 2015
- PPARγ activation ameliorates postoperative cognitive decline probably through suppressing hippocampal neuroinflammation in aged mice. – PubMed – NCBI – 2017
- Postoperative cognitive dysfunction in the aged: the collision of neuroinflammaging with perioperative neuroinflammation. – PubMed – NCBI – 2018
- The Role of Neuroinflammation in Postoperative Cognitive Dysfunction – Moving From Hypothesis to Treatment – 2018
- Treating inflammation and infection in the 21st century: new hints from decoding resolution mediators and mechanisms – 2017
- Structural Elucidation and Physiologic Functions of Specialized Pro-Resolving Mediators and Their Receptors – 2017
- LPS is a Switch for Inflammation in the Gut and Beyond
- Identification of specialized pro-resolving mediator clusters from healthy adults after intravenous low-dose endotoxin and omega-3 supplementation – a methodological validation – 2018
- The Protectin Family of Specialized Pro-resolving Mediators – Potent Immunoresolvents Enabling Innovative Approaches to Target Obesity and Diabetes – 2018
- Protectins and Maresins – New Pro-Resolving Families of Mediators in Acute Inflammation and Resolution Bioactive Metabolome – 2014
- Functional Metabolomics Reveals Novel Active Products in the DHA Metabolome – 2012
Acute Post-Injury Pain – Acetaminophen (Tylenol)
- AM404, an inhibitor of anandamide uptake, prevents pain behaviour and modulates cytokine and apoptotic pathways in a rat model of neuropathic pain – 2006
- AM404, paracetamol metabolite, prevents prostaglandin synthesis in activated microglia by inhibiting COX activity – 2017
- First evidence of the conversion of paracetamol to AM404 in human cerebrospinal fluid – 2017
- Pharmacological Management of Adults with Chronic Non-Cancer Pain in General Practice – 2020
Preventing Pain Related to Surgery – Alpha-2 Agonists
Preventing Pain Related to Surgery – Buprenorphine
- Role of buprenorphine in acute postoperative pain – 2016
- Efficacy and Safety of Transdermal Buprenorphine versus Oral Tramadol:Acetaminophen in Patients with Persistent Postoperative Pain after Spinal Surgery – 2017
- Low-dose buprenorphine infusion to prevent postoperative hyperalgesia in patients undergoing major lung surgery and remifentanil infusion a double-blind, randomized, active-controlled trial – 2017
Preventing Pain Related to Surgery – Diet
Preventing Pain Related to Surgery – Genetics
- genotyping-test-with-clinical-factors-better-management-of-acute-postoperative-pain-2015
- the-impact-of-genetic-variation-on-sensitivity-to-opioid-analgesics-in-patients-with-postoperative-pain-a-systematic-review-and-meta-analysis-2015
- Chronic postsurgical pain – is there a possible genetic link? – 2017
Preventing Pain Related to Surgery – Epigenetics
- Epigenetic-regulation-of-spinal-cord-gene-expression-controls-opioid-induced-hyperalgesia-2014
- Epigenetic-regulation-of-opioid-induced-hyperalgesia-dependence-and-tolerance-in-mice-2013
- Epigenetic-regulation-of-persistent-pain-2015
- Chronic-opioid-use-is-associated-with-increased-dna-methylation-correlating-with-increased-clinical-pain-pubmed-ncbi
- Could targeting epigenetic processes relieve chronic pain states? – PubMed – NCBI
- Epigenetic-mechanisms-of-chronic-pain-2015
- Telomeres and epigenetics – Potential relevance to chronic pain – 2012
- Epigenetics of chronic pain after thoracic surgery. – PubMed – NCBI
- Epigenetics-in-the-perioperative-period-2015
- Epigenetic modulation of mGlu2 receptors by histone deacetylase inhibitors in the treatment of inflammatory pain – PubMed – 2009
- Preventing chronic pain following acute pain – risk factors, preventive strategies, and their efficacy – 2011
Preventing Pain Related to Surgery – Gabapentin & Pregabalin
- Do surgical patients benefit from perioperative gabapentin:pregabalin? A systematic review of efficacy and safety. – PubMed – NCBI
- Perioperative administration of gabapentin 1,200 mg day−1 and pregabalin 300 mg day−1 for pain following lumbar laminectomy and discectomy – 2011
- Preemptive use of gabapentin in abdominal hysterectomy: a systematic review and meta-analysis. – PubMed – NCBI
- The Effect of Gabapentin on Acute Postoperative Pain in Patients Undergoing Total Knee Arthroplasty – 2016
- The prevention of chronic postsurgical pain using gabapentin and pregabalin: a combined systematic review and meta-analysis. – PubMed – NCBI
- The use of gabapentin in the management of postoperative pain after total hip arthroplasty – 2016
- The use of gabapentin in the management of postoperative pain after total knee arthroplasty – 2016
- Use of gabapentin for perioperative pain control – A meta-analysis – 2007
- The efficacy of gabapentin:pregabalin in improving pain after tonsillectomy: A meta-analysis. – PubMed – NCBI
- Effects of gabapentin on postoperative pain, nausea and vomiting after abdominal hysterectomy: a double blind randomized clinical trial. – PubMed – NCBI
- Gabapentin and postoperative pain – a systematic review of randomized controlled trials 2006 – PubMed Health
- Preoperative Preemptive Drug Administration for Acute Postoperative Pain – A Systematic Review And Meta-Analysis – 2016
- the-effects-of-preoperative-oral-pregabalin-and-perioperative-intravenous-lidocaine-infusion-on-postoperative-morphine-requirement-in-patients-undergoing-laparatomy-2015
- perioperative-pain-management- 2007 pubmed-ncbi
- optimizing-pain-management-to-facilitate-enhanced-recovery-after-surgery-pathways 2015 -pubmed-ncbi
- Treatment_of_Neuropathic_Pain_The_Role_of_Unique_Opioid_Agents_-_2016
- The Anti-Allodynic Gabapentinoids – Myths, Paradoxes, and Acute Effects – 2016
- Gabapentinoids as a Part of Multi-modal Drug Regime for Pain Relief following Laproscopic Cholecystectomy: A Randomized Study – 2017
- Effects of pregabalin and gabapentin on postoperative pain and opioid consumption after laparoscopic cholecystectomy – 2017
- Do surgical patients benefit from perioperative gabapentin:pregabalin? A systematic review of efficacy and safety. – PubMed – NCBI
- Impact of pregabalin on acute and persistent postoperative pain: a systematic review and meta-analysis. – PubMed – NCBI
- Effects of pregabalin and gabapentin on postoperative pain and opioid consumption after laparoscopic cholecystectomy – 2017
- Effect of Perioperative Gabapentin on Postoperative Pain Resolution and Opioid Cessation in a Mixed Surgical Cohort – 2018
Preventing Pain Related to Surgery – Ketamine & NMDA Antagonists
- Role of Ketamine in Acute Postoperative Pain Management – A Narrative Review – 2015
- Perioperative ketamine for acute postoperative pain. – PubMed – NCBI
- Ketamine decreases postoperative pain scores in patients taking opioids for chronic pain: results of a prospective, randomized, double-blind study. – PubMed – NCBI
- Intraoperative ketamine reduces perioperative opiate consumption in opiate-dependent patients with chronic back pain undergoing back surgery. – PubMed – NCBI
- Ketamine as an Adjunct to Postoperative Pain Management in Opioid Tolerant Patients After Spinal Fusions – A Prospective Randomized Trial – 2007
- The efficacy of N-methyl-D-aspartate receptor antagonists on improving the postoperative pain intensity and satisfaction after remifentanil-based a… – PubMed – NCBI
- The clinical role of NMDA receptor antagonists for the treatment of postoperative pain. – PubMed – NCBI
- Ketamine – an old drug revitalized in pain medicine – 2017
- Effect of ketamine combined with magnesium sulfate in neuropathic pain patients (KETAPAIN) – study protocol for a randomized controlled trial – 2017
- A systematic review and meta-analysis of ketamine for the prevention of persistent post-surgical pain. – PubMed – NCBI – 2014
- Ketamine for pain – 2017
- Ketamine for pain management – 2018
- Butorphanol and Ketamine Combined in Infusion Solutions for Patient-Controlled Analgesia Administration – A Long-Term Stability Study – 2015
- Multimodal Analgesia, Current Concepts, and Acute Pain Considerations. – PubMed – NCBI -2017
Preventing Pain Related to Surgery – Lidocaine
- Building a diagnostic algorithm on localized neuropathic pain (Lnp) and targeted topical treatment- focus on 5% lidocaine-medicated plaster – 2014
- Peripheral input and its importance for central sensitization – PubMed – 2013
- Treatment of localized post-traumatic neuropathic pain in scars with 5% lidocaine medicated plaster – 2010
- Lidocaine Infusion- A Promising Therapeutic Approach for Chronic Pain – 2017
- Durability of Benefit From Repeated Intravenous Lidocaine Infusions in Fibromyalgia Patients A Case Series and Literature Review – PMC – 2015
- Intravenous lidocaine for fibromyalgia syndrome an open trial – PubMed – 2009
Preventing Pain Related to Surgery – Magnesium
- Perioperative systemic magnesium to minimize postoperative pain: a meta-analysis of randomized controlled trials. – PubMed – NCBI
- Peri-operative intravenous administration of magnesium sulphate and postoperative pain – a meta-analysis – 2013
- Effect of magnesium sulfate on morphine activity retention to control pain after herniorrhaphy. – PubMed – NCBI
- Effect of ketamine combined with magnesium sulfate in neuropathic pain patients (KETAPAIN) – study protocol for a randomized controlled trial – 2017
Preventing Pain Related to Surgery – NSAIDs
See: NSAIDs
- Sulfonamide cross-reactivity: is there evidence to support broad cross-allergenicity? – PubMed – NCBI – 2013
- Should celecoxib be contraindicated in patients who are allergic to sulfonamides? Revisiting the meaning of ‘sulfa’ allergy. – PubMed – NCBI 2001
Preventing Pain Related to Surgery – Nefopam
- nefopam-after-total-hip-arthroplasty-role-in-multimodal-analgesia-pubmed-ncbi
- nefopam-analgesia-and-its-role-in-multimodal-analgesia-a-review-of-preclinical-and-clinical-studies-pubmed-ncbi
- preventive-analgesic-efficacy-of-nefopam-in-acute-and-chronic-pain-after-breast-cancer-surgery-2016
- neuronal-sensitization-and-its-behavioral-correlates-in-a-rat-model-of-neuropathy-are-prevented-by-a-cyclic-analog-of-orphenadrine-pubmed-ncbi
Preventing Pain Related to Surgery – Tapentadol (Nucynta)
Preventing Pain Related to Surgery – Vitamin C
Chronic Regional Pain Syndrome (CRPS)/Reflex Sympathetic Dystrophy (RSD)
- Efficacy of vitamin C in preventing complex regional pain syndrome after wrist fracture – A systematic review and meta-analysis – 2017
- [Vitamin C and prevention of reflex sympathetic dystrophy following surgical management of distal radius fractures]. – PubMed – NCBI
- Complex regional pain syndrome – recent updates – 2013
- Give vitamin C to avert lingering pain after fracture – 2008
- Effect of vitamin C on frequency of reflex sympathetic dystrophy in wrist fractures – a randomised trial – 1999
- Effect of vitamin C on prevention of complex regional pain syndrome type I in foot and ankle surgery. – PubMed – NCBI – 2009
Preventing Pain Related to Surgery – Vitamin C
Lumbar Surgery
Preventing Pain Related to Surgery – Vitamin D
Opioids – Transitioning from Short Term to Long Term Use
- How acute pain leads to chronic opioid use – 2018
- Characteristics of initial prescription episodes and likelihood of long-term opioid use – United States, 2006-2015
- New Persistent Opioid Use After Minor and Major Surgical Procedures in US Adults – 2017
Emphasis on Education
Accurate Clinic promotes patient education as the foundation of it’s medical care. In Dr. Ehlenberger’s integrative approach to patient care, including conventional and complementary and alternative medical (CAM) treatments, he may encourage or provide advice about the use of supplements. However, the specifics of choice of supplement, dosing and duration of treatment should be individualized through discussion with Dr. Ehlenberger. The following information and reference articles are presented to provide the reader with some of the latest research to facilitate evidence-based, informed decisions regarding the use of conventional as well as CAM treatments.
For medical-legal reasons, access to these links is limited to patients enrolled in an Accurate Clinic medical program.
Please note also, that many of the benefits for medications described on this web site include “off-label” use for a medication. Off-label prescribing refers to the use of medication for a condition not named in its FDA approval. Physicians are free to prescribe any medication they want, as long as there is some evidence for usefulness. And remember that the lack of an FDA indication does not necessarily mean lack of efficacy—it sometimes means that no drug company has deemed the investment in clinical trials worth the eventual pay off.
Should you wish more information regarding any of the subjects listed – or not listed – here, please contact Dr. Ehlenberger. He has literally thousands of published articles to share on hundreds of topics associated with pain management, weight loss, nutrition, addiction recovery and emergency medicine. It would take years for you to read them, as it did him.
For more information, please contact Accurate Clinic.
Supplements recommended by Dr. Ehlenberger may be purchased commercially online or at Accurate Clinic.
Please read about our statement regarding the sale of products recommended by Dr. Ehlenberger.
Accurate Supplement Prices