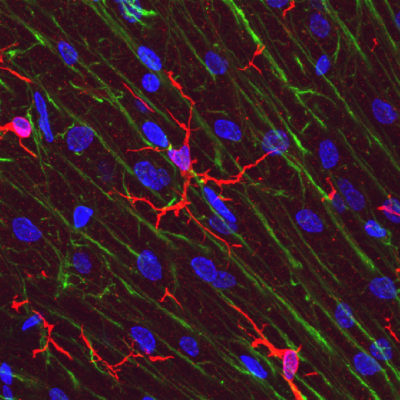
Microglial cells (red) in rat brain
Inflammation
Neuroinflammation
Neuroinflammation is inflammation of nervous tissue in the peripheral or central nervous system. It may occur in response to a variety of triggers including trauma, infection, toxins, or auto-immune processes.
Neuroinflammation plays a central role in chronic pain but also in disorders such as fibromyalgia, depression, PTSD, multiple sclerosis, Parkinson’s Disease, Alzheimer Disease, brain and spinal cord injuries including chronic traumatic encephalopathy (CTE), stroke and schizophrenia.
See:
- Resolving Neuroinflammation
- Systemic Inflammation
- Central Sensitization
- Oxidative Stress, Pain and Disease
- Antioxidants and Oxidative Stress
- Mitochondrial Dysfunction
See also:
- Pain
- Neurobiology of Pain
- Neuropathic (Nerve) Pain
- Neurobiology of Opioids
- Opioids
- Opioid Tolerances
- Medications for Pain
- Gabapentin (Neurontin) & Pregabalin (Lyrica)
- Palmitoylethanolamide (PEA)
- Toll-Like Receptor Antagonists (TLR-4)
- Traumatic Brain Injury
Key to Links:
- Grey text – handout
- Red text – another page on this website
- Blue text – Journal publication
Working on this page, it is still incomplete
Neuroinflammation
Inflammation is a complex biological response that is fundamental to how the body addresses injury and infection to eliminate or resolve the initial cause of cell injury and repair tissues. While acute inflammation is normally a beneficial response directed at healing, chronic inflammation often results from an inappropriate immune response that can lead to tissue damage and ultimately tissue destruction.
Inflammation in the nervous system or “neuroinflammation,” especially when prolonged, can be particularly harmful. While inflammation per se may not cause disease, it contributes importantly to the process of disease in both the peripheral and central nervous systems. Treatment of neuroinflammation may significantly impact the progression and symptomatic manifestation of those conditions associated with neuroinflammation.
Neuroinflammation is implicated in:
- Pain
- Opioid tolerance
- Fibromyalgia
- Reward Deficiency Syndrome (RDS)
- Traumatic brain injury (TBI)
- Arachnoiditis
- Depression
- Multiple Sclerosis
- Alzheimer disease
- Parkinson disease
- Autism Spectrum Disorder
(1). Pain and Neuroinflammation
Initially, acute pain resulting from tissue damage is a means of adaptive survival that allow recognition of injury as a protective mechanism. This acute pain is driven by stimulation of pain receptors associated with tissue injury and associated local inflammation, a normal process of the immune system that facilitates healing. Normally, this acute pain gradually diminishes as the local inflammation resolves and the damaged tissues heal.
In some cases, despite healing of tissues, acute pain progresses to chronic pain, defined generally as pain of greater than 3 months duration. The transition of acute to chronic pain is driven by the failure of acute inflammation involving the nervous system to resolve, a condition termed neuroinflammation, While it is not fully understood why neuroinflammation does not resolve in these cases, the mechanisms of the persisting neuroinflammation involves non-nerve, immune cells in the nervous system.
Neuroinflammation is characterized by the activation of certain immune cells, glial cells such as microglia and astrocytes in the spinal cord and brain. This activation leads to the release of proinflammatory agents called cytokines and chemokines, such as interleukin (IL)-1β and
tumour necrosis factor alpha (TNFα). These compounds impact nerves, which can caused enhanced pain (hyperalgesia and allodynia). Their sustained release in the central nervous system promotes chronic widespread pain that can affect multiple sites throughout the body.
The experience of chronic pain is augmented in some cases by “central sensitization,” a process involving cellular adaptations at nerve synapses along with increased responsiveness to painful stimulation by pain receptors and nerve pathways in the peripheral and central nervous system. This process is driven by neuroinflammation in the peripheral and central nervous system. Central sensitization results in a magnified experience of pain in which the severity of pain becomes greater over time. Furthermore, this magnification of experience is not limited to pain but also involves the experience of other noxious stimuli including touch or foul smells and can lead to impaired tolerance of bright light and loud sounds. Central sensitization can also lead to greater anxiety and emotional irritability.
The understanding of how neuroinflammation drives widespread chronic pain and central sensitization has gained recent attention for its importance in understanding and treating chronic pain and other conditions.
(2). Opioid Tolerance and Neuroinflammation
While opioids can be very effective in treating acute and chronic pain, in many cases their analgesic effectiveness wanes over time leading to less and less benefit that drives the need for higher doses to maintain effectiveness. The loss of effectiveness is termed “opioid analgesic tolerance” (OAT), often simply referred to as “tolerance.”
Classical neuron-centered concepts about tolerance, such as internalization of opioid receptors, upregulation of N-methyl-D-aspartate (NMDA) receptor function, or downregulation of glutamate transporter activity only partially explain the phenomenon of tolerance. Recent evidence confirms that glial activation and upregulation of inflammatory mediators in the central nervous system play pivotal roles in neuropathic pain and opioid tolerance.
(3). Fibromyalgia and Neuroinflammation
Coming soon…
(4). Reward Deficiency Syndrome and Neuroinflammation
See: Reward Deficiency Syndrome and Chronic Pain
(5). Traumatic Brain Injury and Neuroinflammation
(6). Arachnoiditis
See: Arachnoiditis
(7). Depression and Neuroinflammation
Coming soon…
(8). Multiple Sclerosis and Neuroinflammation
Coming soon…
(9). Alzheimer Disease and Neuroinflammation
Neuroinflammation-related microglial activation has been proposed as a mechanism that induces the development of Alzheimer’s disease (AD). Pro-inflammatory activation contributes to AD via decreased glial cell removal of amyloid plaque, the abnormal accumulation of amyloid protein (Aβ) in the brain, a frequent feature of the aging brain and, when excessive, a diagnostic marker of AD.
Neuroinflammation also contributes to increased formation of tau, a protein that polymerizes and tangles nerves – the second diagnostic marker of AD. Thus, the two lesions that characterize AD pathologically – plaques and tangles – arise from two different proteins – Aβ and tau – both of which can misfold and self-assemble into amyloid and both of which are driven by neuroinflammation.
Additional sequela of neuroinflammation includes loss of synaptic function, and cytokine-induced neuron death. Chronic activation of microglia contributes to neurotoxicity by inducing synapse loss that promotes cognitive decline.
Alzheimer Disease and Chronic Pain
Evidence also supports a link between chronic pain and AD based on two interlinked processes, neuroinflammation and noradrenaline (norepinephrine – NE) system dysfunction in the brain areas involved in the emotional components of pain and cognition (the locus coeruleus (LC), prefrontal cortex (PFC) and hippocampus).
The LC is involved in functions including attention, memory, emotion, stress reactions, and pain modification. The LC is the primary source in the brain for manufacturing noradrenaline which drives the descending nerve pathways to the spinal cord. These descending nerve pathways are important for modifying pain and are the target of many drugs used in pain management including duloxetine (Cymbalta) and gabapentin (Neurontin).
A loss of LC-noradrenergic neurons is found in autopsy specimens of AD patients, with 50% to 60% LC cell loss, even more dramatic than the 41% neuron loss observed in the PFC.
Chronic pain induces pathological activation of the locus coeruleus (LC) resulting in an increase of NE release in the prefrontal cortex and hippocampus that triggers microglial pro-inflammatory activation in these areas of the brain. Pro-inflammatory activation may contribute to AD via decreased Aβ phagocytosis, increased tau seeding, loss of synaptic function, and cytokine-induced neuron death.
Clinical observations indicate that intensity of pain is positively correlated with the degree of cognitive impairment and greater degree of pain-related impairment is associated with a higher probability of developing dementia.
Treating Neuroinflammation
See: Resolving Neuroinflammation
A Closer look at Understanding Neuroinflammation
Understanding communication between the nervous system and the immune system is fundamental to understand neuroinflammation. Immune cell-derived inflammatory molecules regulate host responses to inflammation. Although these molecules can originate from various non-neuronal (non-nerve) cells, their most important sources are immune cells: microglia and mast cells, together with astrocytes and possibly also oligodendrocytes. Understanding neuroinflammation also requires an appreciation that non-neuronal cell—cell interactions, between both glia and mast cells and glia themselves, are an integral part of the inflammation process. Within this context the mast cell occupies a key niche in orchestrating the inflammatory process, from initiation to prolongation.
Normal, optimal inflammatory responses and physiological levels of inflammatory mediators are beneficial and protect the body as they remove unwanted waste materials and repair damaged tissues. As such, the initial, acute inflammatory response is protective, and lipid mediators such as eicosanoids (prostaglandins and leukotrienes produced from the essential fatty acid arachidonic acid) play critical roles in the initial response, with interactions between prostaglandins, leukotrienes and pro-inflammatory cytokines amplifying inflammation.
Normally, these altered and reactive immune cells diminish their activity within 10–14 days after injury and the inflammatory response ceases. However, in some cases, this neuroinflammation continues and becomes chronic, leading to many of the manifestations of nerve pain, or “neuropathic” pain, such as hyperalgesia, allodynia and peripheral and central sensitization, all of which are characterized by a magnification of the pain experience.
The Players in Neuroinflammation
The process of neuroinflammation can be understood on a (1) structural level, including the blood-brain barrier (BBB), on a (2) cellular level including immune cells such as mast cells, microglia, astrocytes and oligodendrocytes or on a (3) chemical level including cytokines, chemokines and others.
Neuroinflammation on a Structural Level
Neuroinflammation and the Blood-Brain Barrier (BBB)
In normal physiological conditions, the blood-brain barrier (BBB) prevents entry of most drugs, chemicals, toxins and peripheral blood cells into the brain and central nervous system. The BBB is an extensive network of endothelial cells (ECs) in brain capillaries together with neurons and glial cells, including microglia, that form a neurovascular unit (NVU). The communication between these cells maintains a proper environment for brain function.
The integrity of the BBB which prevents”inappropriate” molecules from entering the central nervous system and brain is dependent on the maintenance of “tight junctions,” where the cells of the blood vessel interface with adjoining cells. Changes in the interactions between blood vessel endothelium and microglia are associated with a variety of inflammation-related diseases in which BBB permeability is compromised. Evidence indicates that activated microglia modulate expression of tight junctions, which are essential for BBB integrity and function. On the other hand, the endothelium can in turn regulate the state of microglial activation.
Trauma and its associated stress induces a local inflammatory response causing disruption and dysfunction of the BBB increasing its permeability. This results in the infiltration of peripheral immune and inflammatory cells such as neutrophils, monocytes, mast cells (see below), and T cells into the brain. These cells become “activated,” immediately releasing inflammatory proteins called cytokines and chemokines within hours post-injury. These mast cell-derived inflammatory mediators further increase blood brain barrier (BBB) permeability and activate localized brain-based immune glial cells such as microglia and astrocytes (see below).
When activated, microglia and astrocytes increase production of similar inflammatory cytokines. Furthermore, all of these inflammatory mediators increase vascular permeability and increase escape and recruitment of immune and inflammatory cells at the site of injury. When the integrity of the BBB is compromised through inflammation or injury, there is increased permeability of the BBB, allowing for increased introduction of inflammatory chemicals, drugs and toxins to enter the central nervous system (CNS) – spinal cord and brain.
Although loss of BBB integrity is associated with several neuropathological disorders including depression, treatments that improve or stabilise the BBB are scarce. At this time, one focus of treatment of impaired BBB integrity lies in the stabilization of glial cells and mast cells. A 2017 study suggests that dehydroepiandrosterone sulfate (DHEAS) supports the integrity of the BBB. DHEA has shown evidence for benefit in the treatment of depression. Palmitoylethanolamide (PEA) has also demonstrated evidence that it glial cells and mast cells.
The Blood Brain Barrier and the Intestinal Epithelial Barrier (IEB)
A growing body of evidence demonstrates that the integrity of the BBB is linked to the integrity of the intestinal epithelial barrier (IEB), the analogous structure to the BBB in the gut. In turn, the integrity of the IEB is linked to the gut microbiome, the populati0n of microbes in the intestinal tract, Disruption of the IEB leads to a condition called “leaky gut syndrome,” in which a disruption of the tight junctions of the cells lining the gut wall allows for the pathologic invasion of compounds within the gut into the blood and systemic circulation.
These invasive compounds include bacterial products and dietary antigens which trigger an immune response causing the release of pro-inflammatory chemicals. This in turn contributes to the condition of systemic inflammation which is tied into neuroinflammation, and the potential development of a number of disease states.
The gut microbiome appears to be a significant factor contributing to the maintenance or the breakdown of the IEB. The gut microbiome is influenced and modified by a number of factors including stress and drugs, in particular NSAIDs and opioids. “Leaky Gut Syndrome” is believed to be associated with many pathological states, especially stress-related disorders. Leaky Gut Syndrome has been implicated in Irritable Bowel Syndrome (IBS), inflammatory bowel disease (Crohn’s and ulcerative colitis), fibromyalgia, depression, headaches and other chronic pain-related conditions.
See: Leaky Gut (Coming soon… )
Neuroinflammation on a Cellular Level
Glial Cells and Neuroinflammation
Glial cells are cells found in the central and peripheral nervous system. They function to maintain balance in nerve and neurotransmitter activity, they form myelin (the coating of some nerve cells), and provide support and protection for neurons (nerve cells). Glial cells are derived from the immune system, the most common of which are microglia cells and astrocytes. At rest, glia cells provide a supportive matrix for nerve cells, supplying nutrients and oxygen and aid in the repair of damaged cells and clear debris to maintain normal brain function.
However, when activated, glial cells are also important in the evolution and maintenance of chronic nerve pain through the release of peptides known as cytokines that may be pro-inflammatory, triggering chronic pain. Sustained activation of pro-inflammatory microglia can lead to a state of chronic neuroinflammation characterized by high concentrations of neurotoxic compounds the brain. Pathologic glial cell activation plays a significant role in the evolution of fibromyalgia pain, central sensitization and other chronic pain syndromes. Glial cells also play a role in opioid function including opioid-induced hyperalgesia and opioid tolerance.
In healthy brains, inflammatory processes normally cease and microglia transition to an anti-inflammatory state. But failure to resolve the pro-inflammatory processes is a characteristic of many neurological disorders.
Following activation, glia cells release pro-inflammatory cytokines/chemokines including:
-
- Tumor necrosis factor (TNF)
- Interleukin-1beta (IL-1β)
- Interleukin-6 (IL-6)
- Interleukin-8 (IL-8)
- Chemokine (C–C motif) ligand 2 (CCL-2), also known as monocyte chemoattractant protein 1 (MCP-1)
- Brain-derived neurotrophic factor (BDNF)
- Nerve growth factor (NGF)
- Glutamate
- Substance P (SP)
Mast Cells and Neuroinflammation
Glial cells participate in inflammation not only directly, but also in to response to molecular mediators produced by other immune system-derived cells, both blood-borne (dendritic cells, lymphocytes, neutrophils), and tissue-resident (mast cells). Mast cell are an important signaling link between the peripheral immune system and the brain in an inflammatory setting. Due to their widespread tissue presence near blood vessels and surfaces exposed to the environment, mast cells function as environmental “sensors” to communicate physiological and/or immune responses. Mast cells detect and respond to changes in environmental temperature and barometric pressure and are believed to play a role in the increased perception of pain associated with changes in weather.
Mast cells are manufactured in the bone marrow and enter the circulation and then into peripheral tissues including connective tissue cells and mucosal cells. They maintain broad tissue distribution, often close to blood vessels and near boundaries between the body’s external environment and the internal milieu, such as skin, mucosa of lungs and digestive tract, and in mouth, eye conjunctiva, and nose. Mast cells also found in the nervous system, including meningeal tissures that surround the brain, brain tissue, and nerve sleeves. They are integral in allergic reactions and anaphylactic shock, stress, mood disorders, inflammatory pain, chronic and neuropathic pain and acute and chronic neurodegenerative disorders.
Mast cells are found in tissues innervated by small caliber sensory nerve fibers (A-delta and C-fibers responsible for pain transmission that extend from the periphery to the spinal cord and brain), in meninges, and apposing cerebral blood vessels. Mast cell’s key role in the inflammatory process, when activated, is to rapidly release granules (degranulation) of bioactive chemicals, pro-inflammatory mediators such as cytokines and others into the surrounding tissues.
Degranulation is triggered by direct injury (physical or chemical), stimulation of immune receptors (such as IgE in allergies) or by activated complement proteins. More than 50 mediators are known and their expression by mast cells is complex and determined to a large extent by tissue location. Additionally, mast cell-derived chemoattractants recruit other immune cells including eosinophils, monocytes, and neutrophils, and can induce T cell activation, proliferation, and cytokine secretion.
It is clear that glial cells and mast cells play major roles in neuroinflammatiom through their release of chemically active protein mediators that impact tissues and stimulate pain, acutely and chronically. Current research is focusing on medications and other agents that can stabilize glial cells and mast cells, suppress their release of mediators and thereby reduce the development and/or maintenance of chronic pain.
Oligodendrocytes and Neuroinflammation
Oligodendrocytes, the myelin-producing cells of the central nervous system (CNS), may also participate in the pain process. In addition to their production of myelin, oligodendrocytes support nerve function and long-term integrity. Oligodendrocyte damage/dysfunction leads to spinal nerve axon pathology and the induction/maintenance of increased pain sensitivity. Also, like glial cells and mast cells, they produce and respond to chemokines/cytokines that modulate CNS immune responses and interact with microglia. In the case of multiple sclerosis (MS), for example, autoimmune inflammation driven by invading peripheral immune cells leads to injury/degeneration of oligodendrocytes and neurons, and contributes to the neuropathic pain often experienced by MS patients.
New Frontier – Resolving Inflammation
The resolution of neuroinflammation has previously been considered a passive process. Recent research, however, has identified mediators with the capacity to actively resolve inflammation, endogenous agents called resolvins, protectins & maresins, that are involved with the process of shutting down neuroinflammation. It is hoped that in the future the means of harnessing these agents for therapeutic purposes will become available.
What is believed at this time, however, is that production of these agents may be promoted by low dose aspirin and omega-3 essential fatty acids while NSAIDs may inhibit their production.
See: Resolving Neuroinflammation
References:
- Introduction to Pain Pathways and Mechanisms
- Pain and the Neuromatrix in the Brain 2001
- Neuropathic pain – mechanisms and their clinical implications – 2014
- Hypothesizing that brain reward circuitry genes are genetic antecedents of pain sensitivity and critical diagnostic and pharmacogenomic treatment targets for chronic pain conditions – 2009
Neuroinflammation – Blood Brain Barrier (BBB)
- Opioids and the Blood-Brain Barrier – A Dynamic Interaction with Consequences on Drug Disposition in Brain – 2017
- Targeting Transporters – Promoting Blood-Brain Barrier Repair in Response to Oxidative Stress Injury – 2015
- The opioid epidemic – a central role for the blood brain barrier in opioid analgesia and abuse – 2017
- The role of the blood–brain barrier in the development and treatment of migraine and other pain disorders – 2014
- Dehydroepiandrosterone sulfate augments blood-brain barrier and tight junction protein expression in brain endothelial cells – 2017
- The impact of microglial activation on blood-brain barrier in brain diseases – 2014
Neuroinflammation – Mast Cells
- Activated mast cells infiltrate in close proximity to enteric nerves in diarrhea-predominant irritable bowel syndrome. – 2003
- Mast cells, glia and neuroinflammation – partners in crime? – 2013
Neuroinflammation – Glial Cells
- evidence-for-brain-glial-activation-in-chronic-pain-patients-2015
- importance-of-glial-activation-in-neuropathic-pain-pubmed-ncbi
- glial-contributions-to-visceral-pain-implications-for-disease-etiology-and-the-female-predominance-of-persistent-pain-2016
- evidence-of-different-mediators-of-central-inflammation-in-dysfunctional-and-inflammatory-pain-interleukin-8-in-fibromyalgia-and-interleukin-1-B-in-rheumatoid-arthritis-2015
- Mast cells, glia and neuroinflammation – partners in crime? – 2013
- Opioid-induced Central Immune Signaling – Implications for Opioid Analgesia – 2015
- Microglia in the spinal cord and neuropathic pain – 2016
- Modulation of microglia can attenuate neuropathic pain symptoms and enhance morphine effectiveness – 2008
- Neuropeptides and Microglial Activation in Inflammation, Pain, and Neurodegenerative Diseases – 2017
- Aβ Plaques -2020
Neuroinflammation – Epigenetics
- the-emerging-field-of-neuroepigenetics-2013
- epigenetic-mechanisms-of-chronic-pain-2015
- epigenetic-regulation-of-chronic-pain-2015
- epigenetic-regulation-of-persistent-pain-2015
- targeting-epigenetic-mechanisms-for-chronic-pain-a-valid-approach-for-the-development-of-novel-therapeutics-pubmed-ncbi
- targeting-epigenetic-mechanisms-for-pain-relief-pubmed-ncbi
- telomeres-and-epigenetics-potential-relevance-to-chronic-pain-2012
- could-targeting-epigenetic-processes-relieve-chronic-pain-states-pubmed-ncbi
Neuroinflammation – Intestinal Epithelial Barrier, Leaky Gut and the Microbiome
- Breaking down the barriers – the gut microbiome, intestinal permeability and stress-related psychiatric disorders – 2015
- Intestinal mucosal barrier function in health and disease – 2009
- Altered intestinal permeability in patients with primary fibromyalgia and in patients with complex regional pain syndrome – 2008
- The Brain-Gut-Microbiome Axis – 2018
- Gut-Brain Psychology – Rethinking Psychology From the Microbiota–Gut–Brain Axis – 2018
- Microbiome—The Missing Link in the Gut-Brain Axis – Focus on Its Role in Gastrointestinal and Mental Health – 2018
- Gut-microbiota-brain axis and effect on neuropsychiatric disorders with suspected immune dysregulation. – 2015
- Microbiome, probiotics and neurodegenerative diseases: deciphering the gut brain axis. – PubMed – NCBI – 2017
- Gut microbiome in health and disease – linking the microbiome- gut-brain axis and environmental factors in the pathogenesis of systemic and neurodegenerative diseases – 2016
- A Review of Traumatic Brain Injury and the Gut Microbiome – Insights into Novel Mechanisms of Secondary Brain Injury and Promising Targets for Neuroprotection – 2018
- The bidirectional gut-brain-microbiota axis as a potential nexus between traumatic brain injury, inflammation, and disease. – PubMed – NCBI
- Microbiome-microglia connections via the gut-brain axis. – PubMed – NCBI
Neuroinflammation – Epigenetics, Acute Pain Transition to Chronic Pain
- epigenetics-and-the-transition-from-acute-to-chronic-pain-2012
- epigenetics-in-
the-perioperative-period-2015 - < a href=”https://accurateclinic.com/wp-content/uploads/2016/03/Epigenetics-of-chronic-pain-after-thoracic-surgery.-PubMed-NCBI-2.pdf”>epigenetics-of-chronic-pain-after-thoracic-surgery-pubmed-ncbi
Neuroinflammation – Epigenetics, Opioids
- chronic-opioid-use-is-associated-with-increased-dna-methylation-correlating-with-increased-clinical-pain-pubmed-ncbi
- epigenetic-regulation-of-opioid-induced-hyperalgesia-dependence-and-tolerance-in-mice-2013
- epigenetic-regulation-of-spinal-cord-gene-expression-controls-opioid-induced-hyperalgesia-2014-no-highlights
Neuroinflammation – Opioid Tolerance
- Opioid-induced Central Immune Signaling – Implications for Opioid Analgesia – 2015
- Microglia in the spinal cord and neuropathic pain – 2016
- Modulation of microglia can attenuate neuropathic pain symptoms and enhance morphine effectiveness – 2008
- Neuroimmune activation and neuroinflammation in chronic pain and opioid tolerance:hyperalgesia. – PubMed – NCBI – 2004
- Role of Neuroinflammation in Opioid Tolerance: Translational Evidence from Human-to-Rodent Studies. – PubMed – NCBI – 2018
Neuroinflammation – Addiction & Reward Deficiency Syndrome (RDS)
Glial and neuroinflammatory targets for treating substance use disorders – 2017
Neuroinflammation – Pain
- Activated mast cells infiltrate in close proximity to enteric nerves in diarrhea-predominant irritable bowel syndrome. – 2003
- An Integrative Approach to Neuroinflammation in Psychiatric disorders and Neuropathic Pain – 2018 Neuroinflammation and Central Sensitization in Chronic and Widespread Pain. – PubMed – NCBI
- A-Pharmacological-Rationale-to-Reduce-the-Incidence-of-Opioid-Induced-Tolerance-and-Hyperalgesia-A-Review-2018
- The Uses of Low-Dose Naltrexone in Clinical Practice
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Alternatives to Opioids in the Pharmacologic Management of Chronic Pain Syndromes – A Narrative Review of Randomized, Controlled, and Blinded Clinical Trials – 2017
Neuroinflammation – Treatment Overviews
- An Integrative Approach to Neuroinflammation in Psychiatric disorders and Neuropathic Pain – 2018 Neuroinflammation and Central Sensitization in Chronic and Widespread Pain. – PubMed – NCBI
- A-Pharmacological-Rationale-to-Reduce-the-Incidence-of-Opioid-Induced-Tolerance-and-Hyperalgesia-A-Review-2018
- The Uses of Low-Dose Naltrexone in Clinical Practice
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Alternatives to Opioids in the Pharmacologic Management of Chronic Pain Syndromes – A Narrative Review of Randomized, Controlled, and Blinded Cli
nical Trials – 2017
Neuroinflammation Treatment – Minocycline
Resolving Inflammation
- Vagus nerve controls resolution and pro-resolving mediators of inflammation – 2014
- The Resolution Code of Acute Inflammation – Novel Pro-Resolving Lipid Mediators in Resolution – 2015
- Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. – PubMed – NCBI – 2018
- Resolvins and protectins – mediating solutions to inflammation – 2009
- Resolvins and inflammatory pain – 2011
- Resolution of inflammation – an integrated view – 2013
- Protectins and maresins – New pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome – 2014
- Proresolving lipid mediators and mechanisms in the resolution of acute inflammation – 2014
- Novel Pro-Resolving Lipid Mediators in Inflammation Are Leads for Resolution Physiology – 2014
- Novel Anti-Inflammatory — Pro-Resolving Mediators and Their Receptors – 2011
- Lipid Mediators in the Resolution of Inflammation – 2015
- PPARγ activation ameliorates postoperative cognitive decline probably through suppressing hippocampal neuroinflammation in aged mice. – PubMed – NCBI – 2017
- Postoperative cognitive dysfunction in the aged: the collision of neuroinflammaging with perioperative neuroinflammation. – PubMed – NCBI – 2018
- The Role of Neuroinflammation in Postoperative Cognitive Dysfunction – Moving From Hypothesis to Treatment – 2018
- Treating inflammation and infection in the 21st century: new hints from decoding resolution mediators and mechanisms – 2017
- Structural Elucidation and Physiologic Functions of Specialized Pro-Resolving Mediators and Their Receptors – 2017
- LPS is a Switch for Inflammation in the Gut and Beyond
- Identification of specialized pro-resolving mediator clusters from healthy adults after intravenous low-dose endotoxin and omega-3 supplementation – a methodological validation – 2018
- The Protectin Family of Specialized Pro-resolving Mediators – Potent Immunoresolvents Enabling Innovative Approaches to Target Obesity and Diabetes – 2018
- Protectins and Maresins – New Pro-Resolving Families of Mediators in Acute Inflammation and Resolution Bioactive Metabolome – 2014
- Functional Metabolomics Reveals Novel Active Products in the DHA Metabolome – 2012
Emphasis on Education
Accurate Clinic promotes patient education as the foundation of it’s medical care. In Dr. Ehlenberger’s integrative approach to patient care, including conventional and complementary and alternative medical (CAM) treatments, he may encourage or provide advice about the use of supplements. However, the specifics of choice of supplement, dosing and duration of treatment should be individualized through discussion with Dr. Ehlenberger. The following information and reference articles are presented to provide the reader with some of the latest research to facilitate evidence-based, informed decisions regarding the use of conventional as well as CAM treatments.
For medical-legal reasons, access to these links is limited to patients enrolled in an Accurate Clinic medical program.
Should you wish more information regarding any of the subjects listed – or not listed – here, please contact Dr. Ehlenberger. He has literally thousands of published articles to share on hundreds of topics associated with pain management, weight loss, nutrition, addiction recovery and emergency medicine. It would take years for you to read them, as it did him.
For more information, please contact Accurate Clinic.
Supplements recommended by Dr. Ehlenberger may be purchase
d commercially online or at Accurate Clinic.
Please read about our statement regarding the sale of products recommended by Dr. Ehlenberger.
Accurate Supplement Prices
.
