Nutriceuticals:
Palmitoylethanolamide (PEA)
Palmitoylethanolamide (PEA), an endogenous (manufactured by the body) fatty acid amide, is emerging as a new agent in the treatment of pain and inflammation. As an endogenous agent and one also found in foods such as eggs and milk, no serious side effects or drug–drug interactions been identified. PEA has been used for many decades in the Netherlands and Europe, but since the 1990s interest in the U.S. has surged. It is classified as a “food for medical purposes” in Europe and as a diet supplement in the U.S.
PEA has demonstrated effectiveness for chronic pain of multiple types associated with many painful conditions, especially with neuropathic (nerve) pain, inflammatory pain and visceral pain such as endometriosis and interstitial cystitis.
The level of confidence (LOC) in the recommendation of PEA as a supplement is high.
Understanding Pain:
- Neurobiology of Pain
- Neuropathic (Nerve) Pain
- Visceral Pain
- Central Sensitization
- Mast Cell Activation Disease (MCAD)
Also:
- Medical Marijuana – Getting Started
- Cannabidiol (CBD) – Drug Actions & Interactions
- Cannabidiol (CBD) – Synergy between CBD and PEA
- Surgical Pain – Post-Operative
Key to Links:
- Grey text – handout
- Red text – another page on this website
- Blue text – Journal publication
Definitions and Terms Related to Pain
Palmitoylethanolamide (PEA)
Brief Summary
LOC: Med-High, based on indication
Palmitoylethanolamide a natural compound manufactured by the body and also found in foods such as eggs and milk PEA is emerging as a new agent in the treatment of pain and inflammation which has been used for many decades in the Netherlands and Europe.Recent interest in PEA ihas surged n the U.S. where itt is classified as a “food for medical purposes.”
Dose: 300-600 mg twice a day, or up to 1200 mg/day
Benefits:
PEA has demonstrated effectiveness for multiple types of chronic pain associated with many conditions, but especially with neuropathic (nerve) pain, inflammatory pain and visceral pain such as endometriosis and interstitial cystitis. Other conditions with evidence of benefit with PEA include:
- Arthritis – osteoarthritis & rheumatoid athritis
- Fibromyalgia
- Peripheral neuropathies – diabetic neuropathy & chemotherapy-induced peripheral neuropathy
- Carpal tunnel syndrome
- Opioid Tolerance and Hyperalgesia
- Low back pain – herniated disc disease, failed back surgery syndrome, other
- Sciatic pain
- Dental pain
- Neuropathic pain related to stroke & multiple sclerosis
- Inflammatory Bowel Disease
- Chronic pelvic pain
- Shingles pain (postherpetic neuralgia)
- Vaginal pain (vulvadynia)
- Post-operative dental surgery pain
- Traumatic Brain Injury/Chronic Traumatic Encephalopathy
Palmitoylethanolamide (PEA) – Overview
Palmitoylethanolamide (PEA), also called palmitoylethanolamine or N-2 hydroxyethyl palmitamide), belongs to the family of N-acylethanolamines (NAEs), naturally occuring, biologically active lipids that act on cannabinoid receptor (CR2) and interact with inflammatory cells in the nervous system.
The history of PEA as a natural food ingredient with medicinal properties was first identified in 1943 as part of an epidemiological study focused on childhood rheumatic fever, which was noted to occur more frequently in those children who ate fewer eggs. Subsequently investigators noted that the occurrence of rheumatic fever was reduced in children fed egg yolk powder. Subsequently PEA was first identified in the 1950s as being an active anti-inflammatory agent in chicken egg yolk.
Conditions with Evidence of Benefit with PEA
- Arthritis – osteoarthritis & rheumatoid athritis
- Fibromyalgia
- Peripheral neuropathies – diabetic neuropathy & chemotherapy-induced peripheral neuropathy
- Carpal tunnel syndrome
- Opioid Tolerance and Hyperalgesia
- Low back pain – herniated disc disease, failed back surgery syndrome, other
- Sciatic pain
- Dental pain
- Neuropathic pain – related to stroke & multiple sclerosis
- Inflammatory Bowel Disease
- Chronic pelvic pain
- Shingles pain (postherpetic neuralgia)
- Vaginal pain (vulvadynia)
- Post-operative dental surgery pain
- Traumatic Brain Injury/Chronic Traumatic Encephalopathy
How does PEA work with pain?
Two particularly interesting processes have been proposed as to how PEA improves pain. First, PEA is not analgesic strictly speaking since it does not modify the physiological pain threshold per se, rather it normalizes conditions of hypersensitivity within the peripheral and central nervous system that arise from neuroinflammatiom. Second, PEA not only relieves pain itself but it also improves pain-induced cognitive impairments through its actions in the brain.
Neuroinflammation
A solid body of evidence growing over the last 5-10 years indicates that chronic pain is largely due to a process called neuroinflammation, a condition characterized by activation of a number of inflammatory cells within the peripheral and central nervous systems. Neuroinflammation is characterized by migration of immune cells into an area of injury which release inflammatory chemical products that lead to activation and maintenance of chronic pain. These inflammatory cells, mast cells and glial cells, are now targets for development of new medications for treating chronic pain. Evidence indicates that suppression of the activation of these cells may limit or abolish the evolution of acute to chronic pain and may also act to reduce chronic pain.
On the forefront of research into agents that may act on neuroinflammation is palmitoylethanolamide (PEA) which has been reported to reduce mast cell activation and to control glial cell behaviors. What is particularly exciting about PEA is that it is a naturally occuring agent produced by the body that has no reported serious side effects or drug-drug interactions, making it an extraordinarily safe treatment option. Over the last few years, especially in Europe and the Netherlands, more and more clinical research and practical experience have confirmed that PEA is an effective treatment option for chronic pain.
PEA is widely distributed in different body tissues, including the nervous system, and is synthesized on demand following stress, injury and/or pain and accumulates in affected tissues with inflammation. PEA serves to reduce inflammation and pain in different chronic pain conditions.
A systematic review article published in 2016 identified all clinical trials conducted between 2010 and 2014 on PEA, including micronized (m.PEA) and ultra-micronized PEA ( u.m.PEA), commercially available forms of PEA structured to improve absorption and activity of PEA. Twelve studies met high standards of research criteria and included 1,188 patients who were treated for chronic pain with m.PEA or u.m.PEA for periods of 21 to 60 days with daily doses ranging from 300 to 1200 mg. The different pain diagnoses included: degenerative conditions in 1,174 patients (failed back surgery, back disorders, carpal tunnel syndrome etc.); neuropathic in 170 patients (brachial plexus injury, diabetic, post-herpetic neuropathies, stroke); and mixed diagnoses in 82 patients (arthritis, cancer and other miscellaneous painful diagnoses).
The results of the study including only the 1,431 patients with initial pain intensity ≥ 4 (on a 1-10 point scale of pain severity) were considered. The study concluded that on average, there was a significant reduction in pain equal to 1 point every 2 weeks for the 2 month study periods. PEA improved pain in all patients regardless of age or gender, although there was a slightly enhanced benefit in male patients under 65 y/o.
Most of the research on PEA has focused on neuropathic (nerve) pain where significant benefits have been identified. But there is a growing body of research indicating that PEA benefits many types of pain besides neuropathic pain which, incidentally, may also be due to the growing appreciation of the role of neuropathic pain in conditions such as arthritis and other inflammatory pain conditions as well as visceral pain syndromes including endometriosis, interstitial cystitis and inflammatory bowel disease.
PEA also appears to possess effectiveness in syndromes associated with chronic pain including depression and anxiety.
See below for more information on mechanisms of action of PEA.
Validity of Research
Regarding the strengths/weaknesses of the available studies on PEA, there are several concerns, mainly the small size of most studies. Studies based on smaller numbers of patients lack statistical strength for their conclusions, leaving the conclusions vulnerable to inaccuracy. Large, well-designed studies are lacking for the most part although the recent meta-analysis in 2016 does support the validity of PEA effectiveness.
Safety and Effectiveness Over Time
PEA is a natural substance produced by the body and found in various foods. It is not an opioid. It is not addictive. Preliminary studies indicate that PEA does not develop pharmacological tolerance or gradually lose effectiveness over time as occurs with opioids. It has been shown to be safe for patients with no reported serious side effects and it is considered to lack acute or chronic toxicity. It does not interfere with other medication therapies nor does it trigger drug-drug interactions. There are no known contraindications for PEA, and patients with reduced kidney and liver function can be treated with PEA, as its metabolism is localized and cellular and independent of kidney and liver functions. As with many medications, the safety with long term use over 60 days has not been well studied although thete are reports in the literature of long term use with no problems identified.
Based on the totality of the evidence reviewed, there is a lack of adverse effects with doses of PEA as high as 1200 mg of microPEA per day. The most common regimen studied was 300 mg twice a day, although a sizeable amount of evidence also supports doses of 1200 mg/day. Adverse side effects have been reported to be absent. In summary, available data from animal and human studies support the safety of PEA in general, and of microPEA specifically, in products intended for human and companion animal consumption.
PEA Products
Formulations of PEA
PEA does not dissolve well in water and as such the rate at which it dissolves in the stomach and intestine is often the rate-limiting step for oral absorption and bioavailability. The rate at which PEA dissolves is influenced by, among other factors, it’s particle size and therefore it is usually micronized or ultra-micronized. (manufactured in very small particle size) in order for it to dissolve more rapidly. As compared to naıve PEA (particle size profile ranging between 100 and 700 lm), micronized and ultramicronized PEA differ in their particles size profile (2–10 lm and 0.8–6 lm at most, respectively). Micronization and ultramicronization processes yield different crystalline structures with higher energy content and smaller particle size which result in better diffusion and distribution of these molecules. Collectively, reaearch findings suggest that micronized or ultramicronized formulations of PEA maximize PEA bioavailability and efficacy.
In addition, a recently released formulation combines PEA with lecithin as a liposomal version of PEA that also allows for a greater absorption rate. Head-to-head comparisons studies of the different formulations of PEA in humans are lacking however, and thus there is no clinical data yet to support the use of one formulation over another.
PEA is currently marketed as a nutraceutical in humans: PeaCure in the U.S.; (NormastTM, PelvilenTM [Epitech]), PeaPureTM [JP Russel Science Ltd]) in some European countries (e.g. Italy, Spain; it is sold as a food supplement in other countries, such as the Netherlands). It also is a constituent of a cream (Physiogel AITM, Stiefel) marketed for dry skin. PeaCure offers a liposomal liquid product and a micronized capsule product whereas PeaPure offers micronized and ultramicronized products.
See: Supplements: PEA (Palmitoylethanolamide)
Topical Formulations
PEA cream for topical applications is available in 2% strength, with recommended application 4x/day as needed.
Vitalitus – Soothamide (Palmitoylethanolamide 2% Topical Cream), 100 ml
Additional ingredients include: immondsia chinensis (Jojoba) Oil, Vitellaria paradoxa (Shea) Butter, Aloe Vera, Palmitoylethanolamide, Boswellia serrata (Indian Frankincense) Resin, Commiphora myrrha (Myrrh)
PEA and Specific Pain Conditions
Fibromyalgia (FM)
A 2015 study evaluated FM patients for a total duration of 3 months in which the patients currently treated with duloxetine (Cymbalta) plus pregabalin (Lyrica) were provided supplementary PEA (PEA-um tablets 600mg twice a day in the first month, and PEA-m tablets 300 mg twice a day in the next 2 months). The addition of PEA was noted to significantly reduce pain scores in the FM patients.
Muscular Cramps
Although the pathophysiology of muscular cramps remains poorly understood, PEA might play a role in stabilizing overactive muscles that give rise to night cramps. While PEA has not been studied extensively, a recent article published in 2016 reports three cases of patients with severe, persistent muscle cramps that responded with complete resolution of the cramps within 2-4 weeks, with PEA dosed at 400mg 2-3x/day.
Arthritis
A growing body of evidence now points to neuroinflammation, locally and more systemically, as a promoter of damage to joints and bones, as well as joint-related functional problems. The disease process underlying joint diseases is currently believed to involve communication between cartilage and the subchondral bone beneath the cartilage in the joint—and a loss of balance between these two structures. Dysregulation of the mast cells in these structures is associated with damage to these structures (cartilage, bone, synovia, matrix, nerve endings, and blood vessels). This process includes neuroinflammation which in turn contributes to the chronic pain associated with arthritis.
Communication between the spinal cord and the joint can cause further neuroinflammatory changes at the spinal level involving the central nervous system and brain. A central sensitization process has also been observed in patients with arthritis, where pain thresholds to pressure and prick stimuli are lower than in healthy subjects,making the person experience pain more easily and severely. This central sensitivity to pain does not correlate with radiological findings, suggesting that central sensitization is the factor that contributes most to arthritis pain.
Unfortunately, current conventional treatment strategies for arthritis are directed only at relieving symptoms and do little to limit progression of the disease process itself.
Recent research has focused on the use of PEA as both an arthritic pain relieving substance but also as a treatment that may slow the progress of further joint deterioration. In synovial (joint) fluid, PEA is normally present at high levels(1,500 pmol/mL), but these levels are markedly reduced in patients with osteoarthritis or rheumatoid arthritis, suggesting a protective role for PEA in these conditions. In experimental models of joint disease, changes in PEA levels were also found in the spinal cord, supporting the theory of dysregulation in PEA metabolism in joint diseases. This suggests that PEA supplementation may prove beneficial in these situations.
Both membrane and nuclear receptors are important targets for controlling arthritis disease progression. Among membrane receptors, endocannabinoids play a key role in bone maintenance. Both cannabinoid receptors CB1 and CB2 are present in the skeleton. CB1 and CB2 receptor agonists have been shown to have a protective role in joint diseases and PEA indirectly acts on the CB2 receptor suggesting potential benefit in arthritis. Agonists at the nuclear receptor PPARc reduce the synthesis of inflammatory agents to prevent breakdown of cartilage and PEA also acts on PPARc receptors.
See: Synergy between CBD and PEA
PEA and Quercetin
Oxidative stress is an important contributing factor in arthritic diseases and the antioxidant quercetin has been used successfully as an adjunct in treatment. Micronized and ultramicronized formulations with better solubility of PEA have been introduced in the Italy and internationally, both in the human and veterinary fields and these formulations show superior activity compared to standard PEA formulations. PEA together with natural polyphenols with antioxidant properties show a synergistic effect with their ability to also target the oxidative stress cascade. A recent new formulation is a co-ultramicronized composite of PEA with quercetin.
An animal study published in 2017 that looked at PEA in combination with quercetin, a natural antioxidant showed that the combination reduced pain and improved joint function, protected cartilage against damage, and reduced levels of inflammatory chemicals in the joint fluid. The magnitude of these benefits was comparable to, or even greater than, those of meloxicam (Mobic), a non-steroidal anti-inflammatory medication similar to ibuprofen.
See: Quercetin
To summarize, PEA shows considerable versatility in counteracting inflammation and offers benefit for arthritis both in regards to reducing the development and maintenance of chronic pain but also to help limit the progress of joint destruction associated with arthritis. There is likely even better benefit when PEA is combination with quercetin, luteolin, curcumin, Vitamin C and/or other natural flavonoid antioxidants.
Traumatic Brain Injury/Chronic Traumatic Encephalopathy (CTE)
Chronic Traumatic Encephalopathy (CTE), is a neurodegenerative disease thought to be associated with a history of repetitive head impacts, such as those sustained through contact sports or military combat. Symptoms of CTE usually manifest years after the head trauma(s). The symptoms of CTE are insidious. They may first manifest as deteriorations in attention, concentration, and memory, as well as disorientation and confusion, and may occasionally be accompanied by dizziness and headaches. Mood conditions may occur such as depression along with irritability, emotional instability, aggressiveness and paranoia. With progression, additional symptoms may develop including a lack of insight and poor judgment which may lead to substance abuse.
PEA’s anti-inflammatory actions involve both neuronal and nonneuronal cells, including microglia and peripheral and central mast cells providing neuroprotective effects including a role in maintaining cellular homeostasis in the face of neuroinflammation, one of the mechanisms involved with the developoment of CTE. Studies in mice show that PEA also protects against amyloid β-peptide-induced learning and memory impairment, another mechanism associated with CTE.
See: Traumatic Brain Injury (TBI)
Post-Operative Pain
Recent studies have indicated that nearly half of all surgical patients still have inadequate pain relief. Multiple mechanisms are involved in postoperative pain including neuroinflammation and mast cell activation. Previous studies have shown that incisions can cause mast cell degranulation resulting in the release of many chemicals that promote the development of acute and chronic pain. Studies suggest that reducing neuroinflammation and stabilizing mast cells reduce post-operative pain,
One study looking at post-operative pain after surgical extraction of impacted lower third molars did show reduction of pain with PEA. While specific studies for post-operative pain are lacking, when one evaluates the mechanisms postulated for the evolution of acute to chronic pain, especially in spinal surgery, the use of PEA as a glial cell inhibitor to reduce the development of pathologic neuroinflammation makes sense. Along with other glial cell inhibitors including resveratrol and low dose naltrexone, PEA may offer a role as a safe supplement to be taken in the post-operative period to reduce the evolution of chronic pain at little risk of side effects or harm.
Pharmacokinetics of PEA
There is very little data available in the open literature concerning the pharmacokinetic properties of PEA. It is not very water soluble, making it slowly absorbed. Different formulations have been made to enhance absorption.
A Closer Look at the Mechanisms of Action of PEA
Particular cells of the immune system intimately associated with or located within the nervous system, i.e., “non-neuronal cells”, are major contributors to the development and maintenance of chronic pain. In particular, mast cells (within the nervous system and in the periphery) and microglia (at spinal and supraspinal (brain) level) interact with neurons under physiological and pathological conditions.
Multiple mechanisms of action of PEA have been identified for PEA, largely focused on it’s effects upon mast cells and glial cells.
PEA acts as an Autacoid Local Injury Antagonist (ALIA), through reducing mast cell de-granulation which releases multiple compounds that trigger inflammation. It has also been found that PEA is synthesized by mast cells and microglia and is able to keep these cells reactivity within normal physiological boundaries, thereby controlling neuroinflammation and chronic pain.
PEA mimics several endocannabinoid-driven activities, although it does not bind to the classical CB receptors. It enhances the physiological activity of cannabinoids by potentiating their affinity for cannabinoid receptors and the orphan G protein-coupled receptor (GPR)-55 receptors. PEA suppresses endocannabinoid metabolic degradation by inhibiting FAAH. PEA may also indirectly enhance the effects of both plant cannabinoids and endocannabinoid as agonists of the transient receptor potential vanilloid type 1 (TRPV1), peroxisome proliferator- activated receptor-α (PPAR-α), and the cannabinoid receptors. PEA also down-regulates mast cells, lessening inflammation. However, PEA activity also involves CB2-like cannabinoid receptors, ATP-sensitive K+-channels, TRP channels, and NFkB.
It has also been shown that PEA not only acts through non-neuronal cells as described above, but may also directly influence neurons. PEA has been shown to (i) exert protective effects on cortical and cerebellar neurons, (ii) control spontaneous GABAergic synaptic activity in striatal neurons, (iii) dose-dependently increase intracellular calcium concentration in sensory neurons thereby desensitizing pain receptors; (iv) modulate the activity of dorsal root ganglion neurons.
Endocannabinoid System
PEA produces indirect receptor-mediated effects within the Endocannabinoid System (ECS), the site of action of endogenous (natural) cannabinoids (or “endocannabinoids”) such as anandamide (AEA), and plant cannabinoids (phytocannabinoids) like CBD and THC. PEA does not directly activate CB1 or CB2 receptors but does indirectly activate these receptors. PEA inhibits FAAH (fatty acid amide hydrolase), the enzyme that breaks down these cannabinoids so it indirectly enhances the activation of CB1 and CB2 receptors by maintaing or elevating endocannabinoid levels, resulting in significant analgesic benefits. Furthermore, PEA directly activates GPR55 receptors.
GPR55 forms receptor heteromer with either CB1 or CB2 receptors raising the possibility that PEA might modulate CB1- and/or CB2-mediated intracellular signaling by targeting the GPR55 protomer in these putative GPR55/CB1 or GPR55/CB2 heterodimers.
Peroxisome Proliferator-Activated Receptor α (PPARα)
PPARs are involved in regulating biological processes such as pain processing, lipid metabolism, energy balance, fat production, inflammation and cell growth and differentiation. When activated PPARs in immune and brain cells regulate gene expression leading to transcription and production of proteins involved in pain processing, fatty acid oxidation and metabolism. PEA produces analgesic effects via PPARα directly and recent studies have demonstrated that PEA increases the levels of CB2 receptor mRNA and protein as a result of PPAR-α activation and indirect activation of CB1 receptors as particularly noted in osteoarthritis pain.
TRPV1 (Transient receptor potential channels, of the vanilloid subtype)
The endocannabinoid, anandamide, is a full agonist of TRPV1, which act as sensory moderators, including pain. They are activated by compounds (ligands), either endogenous (endocannabinoids such as anandamide) or exogenous (capsaicin). They are also triggered by heat, mechanical and osmotic stress. TRPV channels are found in smooth muscle cells, endothelial cells lining blood vessels, as well as in peri-vascular nerves that accompany blood vessels.
PEA increases the TRPV1-mediated effects of the endocannabionoid, anandamide, and capsaicin resulting in reduction of nerve pain such as shingles.
Other Mechanisms
Other PEA targets in pain processing include the orphan GPCR (GPR55) receptor. These are not the only actions of PEA: it also interacts as an agonist with GPR119, another orphan receptor involved in glucagon-like peptide-1 secretion, which may affect endocannabinoid signalling. It has been reported that GPCR activates κ and δ opioid receptors which may be involved in the synergy between PEA, cannabinoids and opioids in pain reduction.
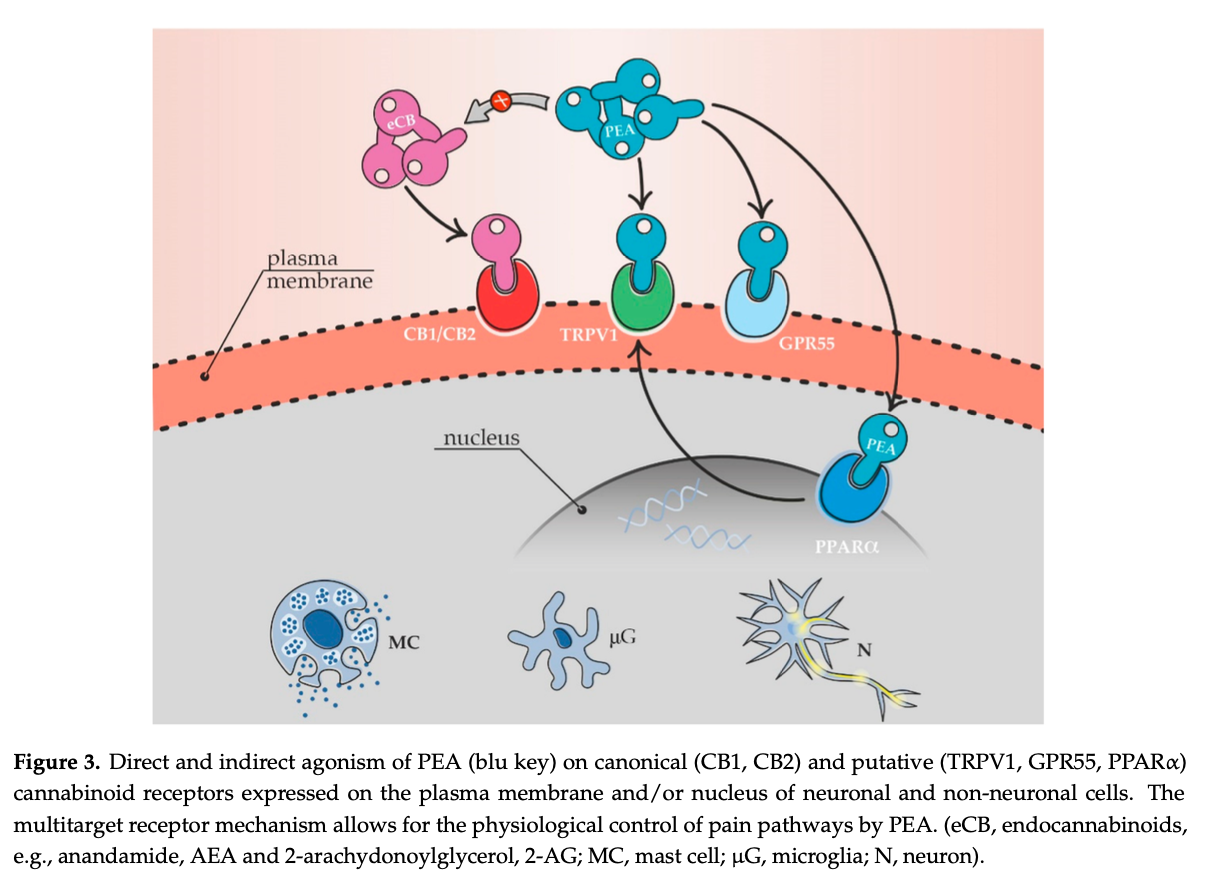
Taken from: “Chronic Pain in Dogs and Cats – Is There Place for Dietary Intervention with Micro-Palmitoylethanolamide?” – 2021
Mast Cells
Mast cells are immune cells mostly located within tissues at the boundary of the external environment, in close proximity to blood vessels and nerve endings and found also within the endoneurial compartment (lining) of peripheral nerves.
Mast cells can modify sensory transmission via a wide spectrum of mediators, including biogenic amines such as histamine and serotonin, cytokines (interleukin-1b (IL-1b) and tumor necrosis factor-a (TNF-a) in particular), enzymes, lipid metabolites, ATP, neuropeptides, nerve growth factor (NGF), and heparin—most of which can interact with sensory nerve terminals. Sensory neurons, in turn, by releasing neuropeptides may provoke mast cell activation/ degranulation.
Mast cell-nerve terminal activity results in nociceptor sensitization, reduced pain threshold at the site of inflammation and, ultimately, dysfunctional pain signaling and hyperalgesia and when it persists, increased responsiveness of nociceptors can also sensitize spinal cord neurons, leading to central sensitization.
Mast Cells and Pain Associated with Cold Weather and Weather Change
Mast cells are perhaps the body’s most sensitive sensors for detecting changes in the external environment. They are located along blood vessels and nerves, particularly near the skin’s surface where they have been shown to be responsive to changes in environmental temperature and baroumetric pressure. It has been proposed (by Dr. Ehlenberger) that given their role in nerve pain and their ability to detect and respond to environmental change, mast cells may play a key role in why patients with chronic pain, especially nerve pain, arthritis and fibromyalgia, experience worse pain in cold weather and with changes in weather associated with changes in barometric pressure as occur prior to weather changes. The stabilizing effect PEA has on mast cells suggests that PEA may be an effective treatment for reducing pain sensitivity to cold weather and weather change.
Glial Cells
Glia cells mediate pain processing at the spinal level. Sensitization of central somatosensory neurons is responsible for the development of chronic neuropathic pain. Microglia, macrophages found in the brain, interact with neurons at the site of injury or disease and can be activated through exposure to a number of molecules, including pro-inflammatory signals released from mast cells. A bidirectional cross talk between brain mast cells and microglia has been theorized, contributing to chronic pain states by releasing pro-inflammatory cytokines, chemokines, and proteases.
Astrocytes, the most abundant glial cell type involved in neuroinflammation, also play a major role in pain processing, contributing to neuropathic pain. When activated, astrocytes release of IL-1b, IL-6, TNF-a and prostaglandin E2. Chronic astrocytic activation in nerve injury results in down-regulation of glutamate transporters, ultimately resulting in decreased glutamate uptake and increased excitatory transmission and facilitation of chronic pain.
PEA, Mast Cells and Glial Cells
These chronic neuroinflammatory processes that sustain neuropathic pain are opposed by the production of lipid mediators that are able to switch off inflammation. Assuming that chronic inflammation may lower the levels or actions of these molecules, it is believed that administration of such lipid mediators might provide an reverse or suppress neuroinflammation. N-acylethanolamines (NAEs) are a class of naturally occurring lipid mediators composed of a fatty acid and ethanolamine—the so-called fatty acid ethanolamines (FAEs). The principal FAEs include the endocannabinoid N-arachidonoylethanolamine (anandamide), and its congeners N-stearoylethanolamine, N-oleoylethanolamine and N-palmitoylethanolamine (PEA or palmitoylethanolamide).
PEA, produced by microglia and mast cells, down-modulates mast cell activation and controls microglial cell activity. By controlling these cells, PEA acts is disease-modifying rather than symptom-modifying, since it acts on the ‘‘roots of the problem’’, i.e., on the cells involved in the generation and maintenance of pain. In support of this theory, PEA levels have been shown to be altered in brain and spinal cord areas involved in pain when pain is induced.
Other Mechanisms of Action
PEA activates peroxisome proliferator activated receptors (PPAR) in cell nuclei of both dorsal root ganglion sensory neurons and glial cells. This receptor is a regulator of gene networks which control pain and inflammation. Stimulation of PPAR-a modulates both the perception and transmission of peripheral pain signaling and spinal amplifying pain mechanisms—thereby exerting its activity in different types and phases of pain. PEA also potentiates the endocannabinoid, anandamide’s, actions at cannabinoid receptors (CB2 receptors) on cell membranes, while itself having no appreciable affinity for either CB1 or CB2 receptors, making it an indirect CB2 agonist.
PEA belongs to a class of lipid autacoids, the N-acylethanolamides. Autacoids are modulating factors that influence the function of cells and tissues which are locally produced on demand and which subsequently are metabolized in the same cells and/or tissues. As an autacoid, PEA is produced in the body on demand and accumulates locally inflammatory and pain disorders.
In addition to its affinity for the PPAR, PEA has high affinity for a number of other targets including the TRPV1 channel that is involved with neuropathic pain and is the site of action of capsaicin, a topical analgesic derived from red peppers. Certain TRPV1 channel activators have been recently patented for the treatment of muscular cramps. PEA can be synthesized in muscle tissue and one of the mechanisms of action for PEA’s anti-cramp activity might be its agonistic action at the TRPV1 receptor. PEA synthesis in muscle tissue seems be disturbed in fibromyalgia. TRPV1 channels are also targets for the cannabinoids, both natural (AEA- or 2-AG) and marijuana-based (CBD and THC). In addition, PEA is also able to increase cannabinoid-induced TRPV1 activation and desensitization .
PEA thus possesses two pain-relieving therapeutic effects, a reduction of neuroinflammation and a reduction in pain signaling and transmission.
Resources:
Purchasing Supplements
To purchase supplements reviewed on this web site or discussed with Dr. Ehlenberger, a discount can be applied to usual commercial pricing by purchasing from either Accurate Clinic’s online Supplement Store or from Carolina Cannabinoids after acquiring the discount code from Accurate Clinic:
- Accurate Clinic’s Supplement Store or, call Toll-Free: 877-846-7122 (Option 1)
- www.CarolinaCannabinoids.US – CBD with terpenes, palmitoylethanolamide (PEA), curcumin and other products.
References
PEA – Availability
Palmitoylethanolamide (PEA)
PEA – New Articles
- Chronic Pain in Dogs and Cats – Is There Place for Dietary Intervention with Micro-Palmitoylethanolamide? – 2021
- Palmitoylethanolamide – A Natural Compound for Health Management – 2021
- Clinical applications of palmitoylethanolamide in pain management – protocol for a scoping review – 2019
- Efficacy of Palmitoylethanolamide for Pain – A Meta-Analysis. – 2017
- evolution-in-pharmacologic-thinking-around-the-natural-analgesic-palmitoylethanolamide-from-nonspecific-resistance-to-PPAR-α-agonist-and-effective-nutraceutical-2013
- Oral Ultramicronized Palmitoylethanolamide – Plasma and Tissue Levels and Spinal Anti-hyperalgesic Effect – 2018
- Palmitoylethanolamide: Problems Regarding Micronization, Ultra-Micronization and Additives – PubMed – 2014
- The Current and Potential Application of Medicinal Cannabis Products in Dentistry – 2021
- The Therapeutic Potential of Cannabis in Counteracting Oxidative Stress and Inflammation – 2021
- Cannabis and Canabidinoids on the Inflammatory Bowel Diseases – Going Beyond Misuse – 2020
- Endocannabinoid System and Its Regulation by Polyunsaturated Fatty Acids and Full Spectrum Hemp Oils – 2021
- CB2 Receptor in Microglia – The Guardian of Self-Control – 2021
- Therapeutic Potential of Cannabidiol (CBD) for Skin Health and Disorders – 2020
- Mast cell–glia axis in neuroinflammation and therapeutic potential of the anandamide congener palmitoylethanolamide – 2012
- The Role of the Brain’s Endocannabinoid System in Pain and Its Modulation by Stress – 2015
- Cannabinoid‐based therapy as a future for joint degeneration. Focus on the role of CB2 receptor in the arthritis progression and pain – an updated review – 2021
- Manipulation of the endocannabinoid system in colitis – A comprehensive review – 2017
- Fibromyalgia – Recent Advances in Diagnosis, Classification, Pharmacotherapy and Alternative Remedies – 2020
- Cannabinoid Receptor-2 Ameliorates Inflammation in Murine Model of Crohn’s Disease – 2017
- Cannabinoids for treating inflammatory bowel diseases – where are we and where do we go? – 2017
- Relevance of Peroxisome Proliferator Activated Receptors in Multitarget Paradigm Associated with theEndocannabinoid System – 2021
- Palmitoylethanolamide and Cannabidiol Prevent Inflammation- induced Hyperpermeability of the Human Gut In Vitro and In Vivo—A Randomized, Placebo-controlled, Double-blind Controlled Trial. – 2018
- Synergistic attenuation of chronic pain using mu opioid and cannabinoid receptor 2 agonists – 2016
- Short-term efficacy of a fixed association of Palmitoylethanolamide and other phytochemicals as add-on therapy in the management of chronic pain in elderly patients – 2018
- Efficacy of a Combination of N-Palmitoylethanolamide, Beta-Caryophyllene, Carnosic Acid, and Myrrh Extract on Chronic Neuropathic Pain – A Preclinical Study – 2019
- Palmitoylethanolamide (PEA) as a Potential Therapeutic Agent in Alzheimer’s Disease – 2019
- Chronic Pain and the Use of Palmitoylethanolamide – An Update – 2018
PEA – Overview
- Information for MDs and Pharmacists on Palmitoylethanolmide
- Palmitoylethanolamide for the treatment of pain – pharmacokinetics, safety and efficacy
- N-palmitoyl-ethanolamine – Biochemistry and new therapeutic opportunities – 2010
- Palmitoylethanolamide in the treatment of chronic pain caused by different etiopathogenesis. 2012 – PubMed – NCBI
- Therapeutic utility of palmitoylethanolamide in the treatment of neuropathic pain associated with various pathological conditions – a case series – 2012
- Professor Rita Levi-Montalcini on Nerve Growth Factor – Mast Cells and Palmitoylethanolamide, an Endogenous Anti-Inflammatory and Analgesic Compound – 2013
- evolution in pharmacologic thinking around the natural analgesic palmitoylethanolamide – from nonspecific resistance to PPAR-α agonist and effective nutraceutical – 2013
- Palmitoylethanolamide – A Natural Body-Own Anti-Inflammatory Agent, Effective and Safe against Influenza and Common Cold – 2013
- Chronic Pain in the Elderly – The Case for New Therapeutic Strategies – 2015
- Palmitoylethanolamide, a Special Food for Medical Purposes, in the Treatment of Chronic Pain – A Pooled Data Meta-analysis – 2016
- Food-Derived Natural Compounds for Pain Relief in Neuropathic Pain – 2016
- Palmitoylethanolamide for the treatment of pain – pharmacokinetics, safety and efficacy – Palmitoylethanolamine for the treatment of pain – 2016
- Palmitoylethanolamide, a Special Food for Medical Purposes, in the Treatment of Chronic Pain – A Pooled Data Meta-analysis – 2016
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Mast cells, glia and neuroinflammation – partners in crime? – 2013
- Efficacy of Palmitoylethanolamide for Pain – A Meta-Analysis. – 2017
- Clinical applications of palmitoylethanolamide in pain management – protocol for a scoping review – 2019
- Chronic Pain in Dogs and Cats – Is There Place for Dietary Intervention with Micro-Palmitoylethanolamide? – 2021
- Palmitoylethanolamide and hemp oil extract exert synergistic anti-nociceptive effects in mouse models of acute and chronic pain – PubMed – 2021
- Palmitoylethanolamide – A Natural Compound for Health Management- 2021
PEA – Anxiety and Depression
PEA – Colds and Flu
- Palmitoylethanolamide – A Natural Body-Own Anti-Inflammatory Agent, Effective and Safe against Influenza and Common Cold – 2013
- Palmitoylethanolamide – A Natural Body-Own Anti-Inflammatory Agent, Effective and Safe against Influenza and Common Cold – 2013
PEA – Pain Conditions/Diagnoses
PEA – Arthritis
- Degenerative Joint Diseases and Neuroinflammation – 2017
- A novel composite formulation of palmitoylethanolamide and quercetin decreases inflammation and relieves pain in inflammatory and osteoarthritic pain models – 2017
- Therapeutic Efficacy of Palmitoylethanolamide and Its New Formulations in Synergy with Different Antioxidant Molecules Present in Diets – 2019
PEA – Back Pain
PEA – Gut and Blood Brain Barriers
- PEA prevented early BBB disruption after cerebral ischaemic_reperfusion (I_R) injury through regulation of ROCK_MLC signaling – PubMed – 2021
- Palmitoylethanolamide and Cannabidiol Prevent Inflammation- induced Hyperpermeability of the Human Gut In Vitro and In Vivo—A Randomized, Placebo-controlled, Double-blind Controlled Trial. – 2018
PEA – Carpal Tunnel and Nerve Compression Syndromes
- Use of palmitoylethanolamide in the entrapment neuropathy of the me… – PubMed – NCBI
- Palmitoylethanolamide, a neutraceutical, in nerve compression syndromes – efficacy and safety in sciatic pain and carpal tunnel syndrome – 2015
- The carpal tunnel syndrome in diabetes – Clinical and electrophysiological improvement after treatment with palmitoylethanolamide. – 2010
- Effect of a new formulation of micronized and ultramicronized N-palmitoylethanolamine in a tibia fracture mouse model of complex regional pain syndrom – 2017
PEA – CRPS (Chronic Regional Pain Syndrome)
- Treatment of chronic regional pain syndrome type 1 with palmitoylethanolamide and topical ketamine cream – 2013
- Effect of a new formulation of micronized and ultramicronized N-palmitoylethanolamine in a tibia fracture mouse model of complex regional pain syndrom – 2017
PEA – Dental Pain
- Randomized Split-Mouth Study on Postoperative Effects of Palmitoylethanolamide for Impacted Lower Third Molar Surgery – 2011
- The Current and Potential Application of Medicinal Cannabis Products in Dentistry – 2021
PEA – Diabetic Neuropathy
PEA – Endometriosis
- Micronized palmitoylethanolamide:trans – polydatin treatment of endometriosis-related pain – A meta-analysis – 2017
- The adjuvant use of N-palmitoylethanolamine and transpolydatin in the treatment of endometriotic pain – 2013
- Effectiveness of the association micronized N-Palmitoylethanolamine (PEA)- transpolydatin in the treatment of chronic pelvic pain related to endometriosis – 2011
PEA – Fibromyalgia
PEA –Headaches (Migraine)
PEA – Multiple Sclerosis
PEA – Muscle Spasm
PEA – Neurodegenerative Diseases (Parkinson’s etc)
PEA – Proctodynia and Vulvodynia (Rectal and Vaginal Pain)
PEA – Post-operative Dental Pain
PEA – Pudendal Neuralgia
PEA – Traumatic Brain Injury (TBI)/Chronic Traumatic Encephalopathy (CTE)
- Palmitoylethanolamide Reduces Neuropsychiatric Behaviors by Restoring Cortical Electrophysiological Activity in a Mouse Model of Mild Traumatic Brain Injury – 2017
- Palmitoylethanolamide is a new possible pharmacological treatment for the inflammation associated with trauma. – PubMed – NCBI
- Molecular evidence for the involvement of PPAR-δ and PPAR-γ in anti-inflammatory and neuroprotective activities of palmitoylethanolamide after spin… – PubMed – NCBI – 2013
- The Nuclear Receptor Peroxisome Proliferator-Activated Receptor-α Mediates the Anti-Inflammatory Actions of Palmitoylethanolamide – 2005
PEA – Pain Types/Mechanisms
PEA – Neurobiology Overviews
- Fatty acid amide hydrolase: biochemistry, pharmacology, and therapeutic possibilities for an enzyme hydrolyzing anandamide, 2-arachidonoylglycerol,… – PubMed – NCBI 2001
- The search for the palmitoylethanolamide receptor – 2005
- Principles of pharmacological research of nutraceuticals 2017
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Mast cells, glia and neuroinflammation – partners in crime? – 2013
PEA – PEA and the Endocannabinoid System
- Effects of homologues and analogues of palmitoylethanolamide upon the inactivation of the endocannabinoid anandamide – 2001
- ‘Entourage’ effects of N-palmitoylethanolamide and N-oleoylethanolamide on vasorelaxation to anandamide occur through TRPV1 receptors – 2008
- Palmitoylethanolamide: from endogenous cannabimimetic substance to innovative medicine for the treatment of cannabis dependence. – PubMed – NCBI – 2013
- Cannabinoids as pharmacotherapies for neuropathic pain – from the bench to the bedside. – 2009
- Cannabinoid-based drugs targeting CB1 and TRPV1, the sympathetic nervous system, and arthritis – 2015
- Palmitoylethanolamide attenuates PTZ-induced seizures through CB1 and CB2 receptors. – PubMed – NCBI – 2015
- Endocannabinoid-related compounds in gastrointestinal diseases – 2018
- Medical Cannabis and Cannabinoids- An Option for the Treatment of Inflammatory Bowel Disease and Cancer of the Colon? – 2018
- Palmitoylethanolamide induces microglia changes associated with increased migration and phagocytic activity – involvement of the CB2 receptor – 2017
- Palmitoylethanolamide, endocannabinoids and related cannabimimetic compounds in protection against tissue inflammation and pain: potential use in c… – PubMed – NCBI
- The ‘Entourage Effect’- How THC can team up with PEA to treat symptoms of Tourette syndrome – 2017
- The ‘Entourage Effect’- How THC can team up with PEA to treat symptoms of Tourette syndrome – 2017
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Cannabinomimetic control of mast cell mediator release: new perspective in chronic inflammation. – PubMed – NCBI
- Palmitoylethanolamide Modulates GPR55 Receptor Signaling in the Ventral Hippocampus to Regulate Mesolimbic Dopamine Activity, Social Interaction, and Memory Processing – 2017
PEA – Fatty Acid Amide Hydrolase (FAAH)
- Fatty acid amide hydrolase: biochemistry, pharmacology, and therapeutic possibilities for an enzyme hydrolyzing anandamide, 2-arachidonoylglycerol,… – PubMed – NCBI 2001
- Fatty Acid Amide Hydrolase – an overview | ScienceDirect Topics – 2009
PEA – PEA and the PPAR
- Molecular evidence for the involvement of PPAR-δ and PPAR-γ in anti-inflammatory and neuroprotective activities of palmitoylethanolamide after spin… – PubMed – NCBI – 2013
- The Nuclear Receptor Peroxisome Proliferator-Activated Receptor-α Mediates the Anti-Inflammatory Actions of Palmitoylethanolamide – 2005
- The Endocannabinoid System and PPARs – Focus on Their Signalling Crosstalk, Action and Transcriptional Regulation – 2021
- Relevance of Peroxisome Proliferator Activated Receptors in Multitarget Paradigm Associated with theEndocannabinoid System – 2021
PEA – Anti-inflammatory Mechanisms
- Palmitoylethanolamide “PEA” – Review of Anti-inflammatory, Analgesic, Neuroprotective Mechanisms
- Palmitoylethanolamide (PEA) – Boosting Its Anti-inflammatory Immune Response
PEA – Central Sensitivity, Oxidative Stress and Pain
- Palmitoylethanolamide in CNS health and disease. – PubMed – NCBI
- Palmitoylethanolamide reduces pain-related behaviors and restores glutamatergic synapses homeostasis in the medial prefrontal cortex of neuropathic mice – 2015
PEA – Mast Cells and Pain
- Mast Cell-Mediated Mechanisms of Nociception – 2015
- Mast Cells in Stress, Pain, Blood-Brain Barrier, Neuroinflammation and Alzheimer’s Disease – 2019
- Mast Cells, Neuroinflammation and Pain in Fibromyalgia Syndrome – 2019
- Mast cell-neural interactions contribute to pain and itch – 2018
- New insights in mast cell modulation by palmitoylethanolamide – PubMed 2013
PEA – Opioid Tolerance and Hyperalgesia
- Delay of morphine tolerance by palmitoylethanolamide
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Ultramicronized N-Palmitoylethanolamine Supplementation for Long-Lasting, Low-Dosed Morphine Antinociception – 2018
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
PEA – Neuroinflammation, Glial Cells and Mast Cells
- Emerging targets in neuroinflammation-driven chronic pain – 2014
- Glia as a Link between Neuroinflammation and Neuropathic Pain – 2012
- Importance of glial activation in neuropathic pain. – PubMed – NCBI
- Mast cells, glia and neuroinflammation – partners in crime? – 2013
- Glia and mast cells as targets for palmitoylethanolamide, an anti-inflammatory and neuroprotective lipid mediator. – PubMed – NCBI
- Mast cell–glia axis in neuroinflammation and therapeutic potential of the anandamide congener palmitoylethanolamide – 2012
- Palmitoylethanolamide induces microglia changes associated with increased migration and phagocytic activity – involvement of the CB2 receptor – 2017
- Palmitoylethanolamide Increases after Focal Cerebral Ischemia and Potentiates Microglial Cell Motility – 2003
- N-Palmitoylethanolamine and Neuroinflammation: a Novel Therapeutic Strategy of Resolution. – PubMed – NCBI
- The palmitoylethanolamide family – a new class of anti-inflammatory agents? – 2002
- Ultra-micronized Palmitoylethanolamide: An Efficacious Adjuvant Therapy for Parkinson’s Disease. – PubMed – NCBI
- An Inflammation-Centric View of Neurological Disease – Beyond the Neuron – 2018
- N-Palmitoylethanolamine and Neuroinflammation: a Novel Therapeutic Strategy of Resolution. – PubMed – NCBI
- A Pharmacological Rationale to Reduce the Incidence of Opioid Induced Tolerance and Hyperalgesia – A Review – 2018
- Mast Cell-Mediated Mechanisms of Nociception – 2015
- Cannabinomimetic control of mast cell mediator release: new perspective in chronic inflammation. – PubMed – NCBI
- Involvement of mast cells in a mouse model of postoperative pain. – PubMed – NCBI – 2011
PEA – Neuropathic Pain
- Microglia in the spinal cord and neuropathic pain – 2016
- Can modulating inflammatory response be a good strategy to treat neuropathic pain? – PubMed – NCBI
- Non-neuronal cell modulation relieves neuropathic pain: efficacy of the endogenous lipid palmitoylethanolamide. – PubMed – NCBI
- Palmitoylethanolamide Is a Disease-Modifying Agent in Peripheral Neuropathy – Pain Relief and Neuroprotection Share a PPAR-Alpha-Mediated Mechanism
- Peroxisome proliferator-activated receptor agonists modulate neuropathic pain – 2014
- delay-of-morphine-tolerance-by-palmitoylethanolamide-2015
- Palmitoylethanolamide, a neutraceutical, in nerve compression syndromes – efficacy and safety in sciatic pain and carpal tunnel syndrome – 2015
- Palmitoylethanolamide in the Treatment of Failed Back Surgery Syndrome – 2017
- Glia and mast cells as targets for palmitoylethanolamide, an anti-inflammatory and neuroprotective lipid mediator. – PubMed – NCBI
- N-Palmitoylethanolamine and Neuroinflammation: a Novel Therapeutic Strategy of Resolution. – PubMed – NCBI
- Palmitoylethanolamide, a Special Food for Medical Purposes, in the Treatment of Chronic Pain – A Pooled Data Meta-analysis – 2016
- Therapeutic utility of palmitoylethanolamide in the treatment of neuropathic pain associated with various pathological conditions – a case series – 2012
- Palmitoylethanolamide, a naturally occurring disease-modifying agent in neuropathic pain. – PubMed – NCBI
- Food-Derived Natural Compounds for Pain Relief in Neuropathic Pain – 2016
- Efficacy of Palmitoylethanolamide for Pain – A Meta-Analysis. – 2017
- Clinical applications of palmitoylethanolamide in pain management – protocol for a scoping review – 2019
- The beneficial use of ultramicronized palmitoylethanolamide as add-on therapy to Tapentadol in the treatment of low back pain – a pilot study – 2017
PEA – Peripheral Neuropathy
- Chronic-idiopathic-axonal-neuropathy-and-pain–treated-with-PEA
- Short-Term Efficacy of Ultramicronized Palmitoylethanolamide in Peripheral Neuropathic Pain
- Micronized Palmitoylethanolamide Reduces the Symptoms of Neuropathic Pain in Diabetic Patients
- Palmitoylethanolamide restores myelinated-fibre function in patient… – PubMed – NCBI
- Short-Term Efficacy of Ultramicronized Palmitoylethanolamide in Peripheral Neuropathic Pain – 2014
- Comment on ‘‘Short-Term Efficacy of Ultramicronized Palmitoylethanolamide in Peripheral Neuropathic Pain’’ – 2014
- Palmitoylethanolamide Reverses Paclitaxel-Induced Allodynia in Mice – 2016
- Topical analgesic creams and nociception in diabetic neuropathy – towards a rationale fundament – 2016
- Treatment of chronic regional pain syndrome type 1 with palmitoylethanolamide and topical ketamine cream – 2013
PEA – Inflammatory Bowel Disease
- Palmitoylethanolamide, a naturally occurring lipid, is an orally effective intestinal anti-inflammatory agent – 2013
- Medical Cannabis and Cannabinoids – An Option for the Treatment of Inflammatory Bowel Disease and Cancer of the Colon? – 2018
PEA – Visceral Pain, Endometriosis
- The adjuvant use of N-palmitoylethanolamine and transpolydatin in the treatment of endometriotic pain – 2013
- Effectiveness of the association micronized N-Palmitoylethanolamine (PEA)- transpolydatin in the treatment of chronic pelvic pain related to endometriosis – 2011
PEA – Trauma
- Palmitoylethanolamide is a new possible pharmacological treatment for the inflammation associated with trauma. – PubMed – NCBI
- Ultramicronized palmitoylethanolamide in spinal cord injury neuropathic pain – A randomized, double-blind, placebo-controlled trial. – 2016
PEA – PEA & Quercetin
PEA – Products
PEA – Liposomal Product Formulations
PEA – Micronization Product Formulations
- A new co-ultramicronized composite including palmitoylethanolamide and luteolin to prevent neuroinflammation in spinal cord injury
- Micronization: a method of improving the bioavailability of poorly soluble drugs. – PubMed – NCBI
- Safety of micronized palmitoylethanolamide (microPEA) – lack of toxicity and genotoxic potential – 2017
- Short-Term Efficacy of Ultramicronized Palmitoylethanolamide in Peripheral Neuropathic Pain – 2014
- Efficacy of ultra-micronized palmitoylethanolamide (um-PEA) in geriatric patients with chronic pain – study protocol for a series of N-of-1 randomized trials – 2015
- Micronized palmitoylethanolamide:trans – polydatin treatment of endometriosis-related pain – A meta-analysis – 2017
- Correction – Effect of a new formulation of micronized and ultramicronized N-palmitoylethanolamine in a tibia fracture mouse model of complex regional pain syndrome – 2018
- Efficacy of ultra-micronized palmitoylethanolamide (um-PEA) in geriatric patients with chronic pain – study protocol for a series of N-of-1 randomized trials – 2016
- Micronized:ultramicronized palmitoylethanolamide displays superior oral efficacy compared to nonmicronized palmitoylethanolamide in a rat model of inflammatory pain – 2014
- Oral Ultramicronized Palmitoylethanolamide – Plasma and Tissue Levels and Spinal Anti-hyperalgesic Effect – 2018
- Safety of micronized palmitoylethanolamide (microPEA) – lack of toxicity and genotoxic potential – 2017
- The pharmacology of palmitoylethanolamide and first data on the therapeutic efficacy of some of its new formulations – 2017
PEA –Safety
- Safety of micronized palmitoylethanolamide (microPEA) – lack of toxicity and genotoxic potential – 2017
- Palmitoylethanolamide for the treatment of pain – pharmacokinetics, safety and efficacy
PEA – Synergy
PEA –Topical
- Topical Analgesics – Critical Issues Related to Formulation and Concentration – 2016
- Topical analgesic creams and nociception in diabetic neuropathy – towards a rationale fundament – 2016
- Treatment of chronic regional pain syndrome type 1 with palmitoylethanolamide and topical ketamine cream – 2013
- Immunomodulatory, Anti-Inflammatory, and Anti-Cancer Properties of Ginseng- A Pharmacological Update – 2023
- Efficacy of a fixed combination of palmitoylethanolamide and acetyl-l-carnitine (PEA+ALC FC) in the treatment of neuropathies secondary to rheumatic diseases – 2021
- Tolerability profile of topical cannabidiol and palmitoylethanolamide a compilation of single-centre randomized evaluator-blinded clinical and in vitro studies in normal skin – PubMed 2021
- Vulvodynia and proctodynia treated with topical baclofen 5 % and palmitoylethanolamide – PubMed 2014
- Topical analgesic creams and nociception in diabetic neuropathy – towards a rationale fundament – 2016Palmitoylethanolamide in the treatment of chronic pain caused by different etiopathogenesis – PubMed – 2012
- Clinical applications of palmitoylethanolamide in pain management- protocol for a scoping review – 2019
- Effects of Palmitoylethanolamide (PEA) on Nociceptive, Musculoskeletal and Neuropathic Pain- Systematic Review and Meta-Analysis of Clinical Evidence – 2022
- Palmitoylethanolamide in the Treatment of Chronic Pain- A Systematic Review and Meta-Analysis of Double-Blind Randomized Controlled Trials – 2023
- Palmitoylethanolamide is a new possible pharmacological treatment for the inflammation associated with trauma – PubMed 2013
- Efficacy of Palmitoylethanolamide for Pain- A Meta-Analysis – 2017
- New Approach to Chronic Back Pain Treatment- A Case Control Study – 2023
- Treatment of chronic regional pain syndrome type 1 with palmitoylethanolamide and topical ketamine cream- modulation of nonneuronal cells – 2013
- Tolerability profile of topical cannabidiol and palmitoylethanolamide a compilation of single-centre randomized evaluator-blinded clinical and in vitro studies in normal skin – PubMed – 2021
- Treatment of chronic regional pain syndrome type 1 with palmitoylethanolamide and topical ketamine cream – modulation of nonneuronal cells – 2013
Topicals – Neuropathic Pain
- Topical Treatments and Their Molecular Cellular Mechanisms in Patients with Peripheral Neuropathic Pain-Narrative Review – 2021
- Novel Drug Targets and Emerging Pharmacotherapies in Neuropathic Pain – 2023
- A pharmacological treatment algorithm for localized neuropathic pain – PubMed 2016
- Management of chronic neuropathic pain with single and compounded topical analgesics – PubMed – 2017
- A Systematic Review and Meta-Analysis on the Role of Nutraceuticals in the Management of Neuropathic Pain in In Vivo Studies – 2022
- Peripheral Mechanisms of Neuropathic Pain-the Role of Neuronal and Non-Neuronal Interactions and Their Implications for Topical Treatment of Neuropathic Pain – 2021
- Topical Treatments for Localized Neuropathic Pain – 2017
- Localized neuropathic pain- an expert consensus on local treatments – 2017
- Treatment of chronic regional pain syndrome type 1 with palmitoylethanolamide and topical ketamine cream – modulation of nonneuronal cells – 2013
- An Insight into Potential Pharmacotherapeutic Agents for Painful Diabetic Neuropathy – 2022
- Topical treatments for diabetic neuropathic pain (Review) – 2019
Topicals – Rheumatoid Arthritis
- Triterpenes as Potential Drug Candidates for Rheumatoid Arthritis Treatment – 2023
- Natural medicines of targeted rheumatoid arthritis and its action mechanism – 2022
- Rheumatiod Arthritis- An Updated Overview of Latest Therapy and Drug Delivery – 2019
Topicals – Sesame Oil
- Topical Anti-Inflammatories- Analgesic Options for Arthritis Beyond NSAIDs – 2021
- Effects of sesame (Sesamum indicum L.) and bioactive compounds (sesamin and sesamolin) on inflammation and atherosclerosis- A review = 2023
- The efficacy of topical sesame oil in patients with knee osteoarthritis A randomized double-blinded active-controlled non-inferiority clinical trial – PubMed – 2019
- The Effects of Topical Sesame (Sesamum indicum) Oil on Pain Severity and Amount of Received Non-Steroid Anti-Inflammatory Drugs in Patients With Upper or Lower Extremities Trauma – 2015
- Effect of light pressure stroking massage with sesame (Sesamum indicum L.) oil on alleviating acute traumatic limbs pain A triple-blind controlled trial in emergency department – PubMed – 2017
Topical Terpenes
Terpenes – Routes of Use:
- The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application – 2020
- Antiviral effect of phytochemicals from medicinal plants – Applications and drug delivery strategies – 2020
- Cannabinoid Delivery Systems for Pain and Inflammation Treatment – 2018
Terpenes – CB2 Receptor
- Cannabinoid-based therapy as a future for joint degeneration. Focus on the role of CB2 receptor in the arthritis progression and pain – an updated review – 2021
- β-Caryophyllene, a CB2 receptor agonist produces multiple behavioral changes relevant to anxiety and depression in mice – 2014
- The CB2 receptor and its role as a regulator of inflammation – 2016
- Cannabinoid CB2 Receptors Regulate Central Sensitization and Pain Responses Associated with Osteoarthritis of the Knee Joint
- A focused review on CB2 receptor-selective pharmacological properties and therapeutic potential of β-caryophyllene, a dietary cannabinoid – 2021
- Topical Terpenes – Wound Healing:
- Beta-caryophyllene enhances wound healing through multiple routes – 2019
Topicals – Terpenes
Topical β-Caryophyllene
Topical Commercial Products
Topical BCP/PEA – Tulla Botanicals, References
- A focused review on CB2 receptor-selective pharmacological properties and therapeutic potential of β-caryophyllene, a dietary cannabinoid – 2021
- Beta Caryophyllene-Loaded Nanostructured Lipid Carriers for Topical Management of Skin Disorders – 2023
- Beta-caryophyllene as an antioxidant, anti-inflammatory and re-epithelialization activities in a rat skin wound excision model – 2022
- Beta-caryophyllene enhances wound healing through multiple routes – 2019
- Beta-caryophyllene-is-a-dietary-cannabinoid-2008
- Efficacy of a Combination of N-Palmitoylethanolamide, Beta-Caryophyllene, Carnosic Acid, and Myrrh Extract on Chronic Neuropathic Pain – A Preclinical Study – 2019
- Hemp Seed Oil in Association with β-Caryophyllene, Myrcene and Ginger Extract as a Nutraceutical Integration in Knee Osteoarthritis – 2022
- Improvement of Oxidative Stress and Mitochondrial Dysfunction by β-Caryophyllene – A Focus on the Nervous System – 2021
- Multi-Target Effects of ß-Caryophyllene and Carnosic Acid at the Crossroads of Mitochondrial Dysfunction and Neurodegeneration- From Oxidative Stress to Microglia-Mediated Neuroinflammation – 2022
- Myrtenal and β-caryophyllene oxide screened from Liquidambaris Fructus suppress NLRP3 inflammasome components in rheumatoid arthritis – 2021
- Non-clinical toxicity of β-caryophyllene, a dietary cannabinoid Absence of adverse effects in female Swiss mice – PubMed – 2018
- Plant Natural Sources of the Endocannabinoid ( E)-β-Caryophyllene- A Systematic Quantitative Analysis of Published Literature – 2020
- Protective Effects of (E)-β-Caryophyllene (BCP) in Chronic Inflammation – 2020
- Protective Effects of (E)-β-Caryophyllene (BCP) in Chronic Inflammation – 2020
- The CB2 Agonist β-Caryophyllene in Male and Female Rats Exposed to a Model of Persistent Inflammatory Pain – 2020
- The Endocannabinoid System, Cannabinoids, and Pain – 2013
- β-Caryophyllene inhibits high glucose-induced oxidative stress, inflammation and extracellular matrix accumulation in mesangial cells – PubMed-2020
- β-Caryophyllene-Loaded Microemulsion-Based Topical Hydrogel- A Promising Carrier to Enhance the Analgesic and Anti-Inflammatory Outcomes – 2023
- β-Caryophyllene, a CB2 receptor agonist produces multiple behavioral changes relevant to anxiety and depression in mice – PubMed – 2014
- β-Caryophyllene, a CB2 Receptor-Selective Phytocannabinoid, Suppresses Motor Paralysis and Neuroinflammation in a Murine Model of Multiple Sclerosis -2017
- β-Caryophyllene, a CB2-Receptor-Selective Phytocannabinoid, Suppresses Mechanical Allodynia in a Mouse Model of Antiretroviral-Induced Neuropathic Pain – 2019
- β-Caryophyllene, A Natural Dietary CB2 Receptor Selective Cannabinoid can be a Candidate to Target the Trinity of Infection, Immunity, and Inflammation in COVID-19 – 2021
- β-Caryophyllene, a Natural Sesquiterpene, Attenuates Neuropathic Pain and Depressive-Like Behavior in Experimental Diabetic Mice – PubMed – 2019
National Academy of Sciences
This website appears to be good resource for exploring medical marijuana.
Emphasis on Education
Accurate Clinic promotes patient education as the foundation of it’s medical care. In Dr. Ehlenberger’s integrative approach to patient care, including conventional and complementary and alternative medical (CAM) treatments, he may encourage or provide advice about the use of supplements. However, the specifics of choice of supplement, dosing and duration of treatment should be individualized through discussion with Dr. Ehlenberger. The following information and reference articles are presented to provide the reader with some of the latest research to facilitate evidence-based, informed decisions regarding the use of conventional as well as CAM treatments.
For medical-legal reasons, access to these links is limited to patients enrolled in an Accurate Clinic medical program.
Should you wish more information regarding any of the subjects listed – or not listed – here, please contact Dr. Ehlenberger. He has literally thousands of published articles to share on hundreds of topics associated with pain management, weight loss, nutrition, addiction recovery and emergency medicine. It would take years for you to read them, as it did him.
For more information, please contact Accurate Clinic.
Supplements recommended by Dr. Ehlenberger may be purchased commercially online or at Accurate Clinic.
Please read about our statement regarding the sale of products recommended by Dr. Ehlenberger.
Accurate Supplement Prices
Emphasis on Education
Accurate Clinic promotes patient education as the foundation of it’s medical care. In Dr. Ehlenberger’s integrative approach to patient care, including conventional and complementary and alternative medical (CAM) treatments, he may encourage or provide advice about the use of supplements. However, the specifics of choice of supplement, dosing and duration of treatment should be individualized through discussion with Dr. Ehlenberger. The following information and reference articles are presented to provide the reader with some of the latest research to facilitate evidence-based, informed decisions regarding the use of conventional as well as CAM treatments.
For medical-legal reasons, access to these links is limited to patients enrolled in an Accurate Clinic medical program.
Should you wish more information regarding any of the subjects listed – or not listed – here, please contact Dr. Ehlenberger. He has literally thousands of published articles to share on hundreds of topics associated with pain management, weight loss, nutrition, addiction recovery and emergency medicine. It would take years for you to read them, as it did him.
For more information, please contact Accurate Clinic.
Supplements recommended by Dr. Ehlenberger may be purchased commercially online or at Accurate Clinic.
Please read about our statement regarding the sale of products recommended by Dr. Ehlenberger.
Accurate Supplement Prices